Content Menu
● How Oxygen Machines Use Water
● Why Distilled Water Is Recommended
● Distilled Water vs Other Water Types
● How Distilled Water Protects the Oxygen Machine
● Patient Safety and Comfort Considerations
● Practical Use: Filling and Changing Distilled Water
● Distillation Water Machine and In‑House Water Preparation
● Integrating Distillation Water Machine into Facility Design
● Choosing a Distillation Water Machine for Oxygen Facilities
● Maintenance and Cleaning of Humidifier Components
● Common Misconceptions About Water Use
● How Everheal Solutions Support Oxygen Systems
● Conclusion
● FAQs About Distilled Water and Oxygen Machines
>> 1. Is distilled water mandatory for every oxygen concentrator?
>> 2. How often should the distilled water be changed?
>> 3. Can I use boiled tap water instead of distilled water?
>> 4. What happens if tap water is used in the oxygen machine humidifier?
>> 5. Why should hospitals and pharma plants install a Distillation Water Machine?
Using distilled water in the humidifier bottle of an oxygen machine (oxygen concentrator) is not only safe but also strongly recommended by most manufacturers and respiratory care providers. Distilled water prevents mineral deposits, reduces microbial growth, and helps protect both the patient and the device during long‑term oxygen therapy.
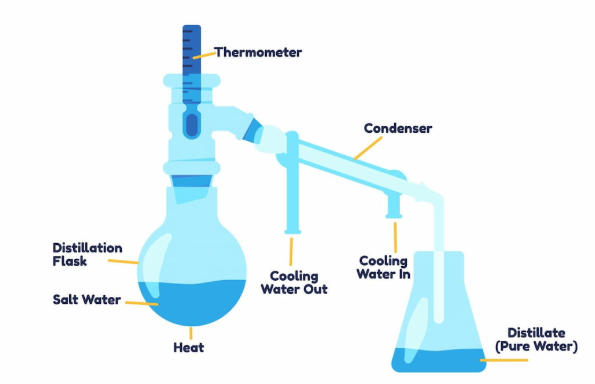
How Oxygen Machines Use Water
An oxygen concentrator takes in room air, removes most of the nitrogen, and delivers a continuous flow of oxygen through a nasal cannula or mask. The oxygen‑generation process uses filters and molecular sieves, not water. However, when oxygen is supplied at higher flow rates or for extended periods, many patients experience dryness of the nose, mouth, and throat.
To solve this problem, many oxygen machines are equipped with a humidifier bottle. The oxygen passes through this bottle, bubbling through water and absorbing moisture before it reaches the patient. This added humidity makes breathing more comfortable, reduces irritation of the mucous membranes, and helps minimize nosebleeds or sore throats caused by very dry oxygen.
Why Distilled Water Is Recommended
Distilled water is produced by heating water to create steam and then condensing that steam back into liquid form. During this distillation process, most minerals, salts, and impurities are left behind, resulting in water that has very low mineral content and very low conductivity. This ultra‑clean profile is precisely why distilled water is recommended for sensitive medical devices.
When distilled water is used in an oxygen machine humidifier, it does not leave behind calcium, magnesium, or other mineral residues on the walls of the bottle or inside narrow passages. That means no limescale, no hard crusts, and far fewer deposits that could block small channels or reduce flow. By contrast, tap water or mineral‑rich bottled water gradually forms scale, which can clog parts of the humidifier and increase maintenance needs.
Another advantage is microbiological safety. Although distillation is not a guarantee of sterility, distilled water typically contains fewer microorganisms than ordinary tap water. When combined with proper cleaning routines, this helps reduce the risk that bacteria or fungi will multiply in the humidifier bottle and be carried into the patient's airways.
Distilled Water vs Other Water Types
Many oxygen therapy users wonder whether they can substitute boiled water, filtered water, or bottled drinking water for distilled water. These options may look similar, but they behave very differently inside an oxygen machine.
Boiled tap water may kill a portion of microorganisms, but it does not remove dissolved minerals. Once that water cools and sits in the humidifier bottle, the minerals are still present and can still form deposits. Filtered water can remove some particles and contaminants, yet common household filters are not designed to fully remove minerals to the level needed by medical equipment.
Bottled drinking water, especially mineral water or “enhanced” water, can actually contain even more dissolved minerals than tap water. Using it in an oxygen machine humidifier accelerates limescale formation. Deionized or reverse osmosis water can be closer to distilled water in terms of purity, but the exact quality depends on the system and maintenance. This is why manufacturers repeatedly specify “distilled water only” in their instructions: it is a clear, standardized recommendation that is easy to follow and minimizes risk.
In medical environments such as hospitals, sterile water is often used in dedicated, closed humidification systems. While sterile water is microbially safer, it may still contain small amounts of minerals depending on how it is produced. In many cases, distilled water or high‑grade purified water is used as the base for sterile water, combining both low mineral content and controlled microbiology.
How Distilled Water Protects the Oxygen Machine
The internal design of an oxygen machine includes fine tubing, valves, connectors, and sometimes sensitive flow‑control components associated with the humidifier path. When hard water is used, minerals precipitate out and adhere to these surfaces. Over time, the deposits narrow the effective cross‑section of the tubing, create rough surfaces that disturb flow, and may even block small orifices.
This scaling can cause several problems. First, it may reduce the level of humidification because oxygen no longer bubbles effectively through the water. Second, it can create irregular back‑pressure on the machine, forcing the compressor to work harder and potentially shortening its lifespan. Third, mineral deposits can complicate cleaning and disinfection, leaving residues that harbor microorganisms or interfere with disinfectants.
Using distilled water prevents most of these issues before they start. By drastically reducing scale formation, distilled water helps maintain stable flow, keeps components cleaner for longer, and reduces the frequency of service calls or part replacements. In the long run, the small cost of regularly purchasing or producing distilled water is far outweighed by the savings in maintenance and the added reliability of the oxygen machine.
Patient Safety and Comfort Considerations
From the patient's perspective, the main reason to use a humidifier is comfort. Dry oxygen can lead to itching, burning, or a feeling of rawness in the nasal passages. Over time, this irritation can trigger nosebleeds, coughing, or increased mucus production as the body tries to protect the mucous membranes.
Distilled water supports safe and comfortable humidification. It provides moisture without introducing additional minerals and contaminants. When used correctly in a clean humidifier bottle, distilled water helps deliver a smooth, gentle airflow that is easier to tolerate during long therapy sessions.
There is also an infection‑control dimension. Water that sits too long in a warm environment can become a breeding ground for bacteria and mold. If that water is then aerosolized through bubbling oxygen, microorganisms can be carried into the patient's respiratory tract. Combining distilled water with proper daily water changes and routine cleaning of the humidifier bottle significantly lowers this risk.
Practical Use: Filling and Changing Distilled Water
Using distilled water properly in an oxygen machine is straightforward but must follow a few important steps:
1. Turn off and unplug the oxygen concentrator before handling the humidifier bottle.
2. Carefully remove the bottle from its holder, taking care not to damage any O‑rings or seals.
3. Pour out any remaining water instead of topping it off. Standing water should never be left for extended periods.
4. Rinse and clean the bottle as recommended in the user manual, using mild detergent or approved disinfectants if specified.
5. Refill the bottle with fresh distilled water up to, but not above, the indicated maximum fill line. Overfilling can lead to water entering the tubing.
6. Reattach the bottle securely, ensuring there are no leaks and that all connections are tight.
Most manufacturers recommend that the water be checked every day and replaced at least once daily or whenever it appears cloudy, discolored, or has any odor. In very dry climates or at high flow rates, the water level may drop quickly; users must never let the bottle run completely dry during operation.

Distillation Water Machine and In‑House Water Preparation
For homes, buying small bottles of distilled water from pharmacies or supermarkets is usually sufficient. However, hospitals, clinics, and pharmaceutical plants often need large and continuous volumes of high‑purity water. In these cases, installing a dedicated Distillation Water Machine becomes a powerful solution.
A Distillation Water Machine produces distilled water on site by heating feedwater to generate steam and then condensing that steam in a controlled, sanitary environment. The equipment can be designed to achieve very low conductivity and low total organic carbon, as required by pharmaceutical and medical guidelines. When correctly specified and validated, a Distillation Water Machine assures a stable supply of distilled water all year round.
For oxygen systems, this means constant availability of high‑quality water for every humidifier bottle in the facility. Instead of relying on delivered containers, staff can draw distilled water directly from a validated distribution loop. This not only improves logistics but also supports traceability and quality control, since the entire system—from feedwater to distillate tank—is monitored and documented.
Integrating Distillation Water Machine into Facility Design
Companies like Everheal, which specialize in pharmaceutical equipment, focus not only on individual machines but also on how those machines fit into a complete factory or hospital layout. A Distillation Water Machine can be integrated with pure water preparation units, pure steam generators, water for injection (WFI) tanks, and sterilization systems. This integration creates a unified water and steam utility backbone.
In a typical design, raw water is first treated through filtration, softening, and reverse osmosis. The pre‑treated water then enters the Distillation Water Machine, which produces distilled water that is either used directly or stored in hygienic tanks. From there, pumps and distribution piping deliver the water to oxygen machine filling stations, autoclaves, cleaning‑in‑place (CIP) systems, and other process users.
Everheal's experience in customized plant layout planning ensures that this network of equipment is arranged efficiently, with minimized dead‑legs, proper slopes for drainage, and optimized access for maintenance. By treating oxygen machine humidification as one element of a wider pharmaceutical water and steam strategy, Everheal helps clients simplify compliance with GMP and other international standards.
Choosing a Distillation Water Machine for Oxygen Facilities
When deciding on a Distillation Water Machine for a hospital or pharmaceutical plant that uses oxygen systems, several key factors must be considered:
- Capacity: The system must supply enough distilled water to cover all uses—oxygen humidifiers, pure steam generators, washing machines, and possible future expansion. Engineers need to calculate peak usage and include a safety margin.
- Water quality: Required limits for conductivity, total organic carbon (TOC), endotoxins, and microbial counts define how the machine and its pre‑treatment must be configured. For pharmaceutical production, these limits are often aligned with major pharmacopoeias.
- Energy efficiency: Distillation consumes energy, so heat‑recovery features, multiple‑effect designs, and good insulation can drastically reduce operational costs.
- Automation and control: A modern Distillation Water Machine usually includes PLC‑based control, alarms, and data logging to ensure reliable operation and help with validation and audits.
- Integration: The unit must connect seamlessly to storage tanks, distribution loops, and monitoring systems used for pure water and pure steam.
Everheal can tailor each Distillation Water Machine to match client specifications, from small clinics that only need modest distilled water output to large pharmaceutical campuses with intensive oxygen therapy and sterile manufacturing processes.
Maintenance and Cleaning of Humidifier Components
Even with the best water quality, routine maintenance of the humidifier bottle and accessories is essential. Distilled water minimizes scale, but dust, skin oils, and environmental contaminants can still find their way into the bottle. If maintenance is neglected, biofilm or residues may gradually build up on interior surfaces.
Manufacturers typically recommend a regular cleaning schedule, which may include daily rinsing and weekly or periodic deep cleaning. Deep cleaning might involve soaking the bottle in a mild detergent solution, thoroughly rinsing with distilled water, and occasionally using approved disinfectants. After cleaning, the bottle should be allowed to air‑dry completely before being refilled when the device is not in active use.
In professional healthcare environments, many facilities choose disposable or single‑patient humidifier bottles that are replaced at fixed intervals. This strategy reduces cross‑contamination risks and simplifies workflows. However, no matter the setting, the guiding principles remain the same: use distilled water, replace it frequently, and keep all components as clean as possible.
Common Misconceptions About Water Use
Users of oxygen machines often encounter conflicting advice from friends, online sources, or local practice. Clarifying common misconceptions helps ensure that both safety and equipment life are maximized.
One widespread myth is that “clear water is safe water.” In reality, water can look crystal clear and still contain high levels of minerals or microorganisms. Another misunderstanding is that if the oxygen machine seems to work normally, the type of water does not matter. Problems caused by mineral buildup or contamination often develop slowly and remain hidden until performance is noticeably affected.
Some users believe that topping off old water is acceptable as long as they are using distilled water. This approach, however, extends the time water remains in the bottle and encourages microbial growth. Best practice is always to completely empty the bottle, clean it if needed, and then refill it with fresh distilled water.
Finally, it is sometimes assumed that distilled water is only necessary in very strict hospital environments. In fact, home oxygen therapy patients can benefit just as much from using distilled water, because they are often on oxygen for many hours each day and rely on their equipment continuously.
How Everheal Solutions Support Oxygen Systems
Everheal is a Chinese company dedicated to advanced pharmaceutical equipment and integrated process solutions. The company's portfolio includes pure water preparation systems, pure steam generators, multifunctional distillation water machines, liquid filling and sealing machines, and various sterilization systems. These technologies are designed to meet the demanding requirements of global pharmaceutical and healthcare clients.
By providing a Distillation Water Machine at the heart of a pure water system, Everheal enables hospitals and pharmaceutical plants to secure a stable, high‑purity water supply for oxygen machine humidifiers and many other critical processes. Combined with custom factory layout planning and turnkey production line construction, Everheal helps customers build safe, efficient, and regulation‑ready facilities.
For clients who need complete solutions, Everheal can integrate distilled water production with oxygen supply, washing processes, autoclaves, and aseptic filling lines. This holistic approach ensures that every step—from the water used to humidify oxygen to the water used in sterilization and product formulation—meets the same high purity standards.
Conclusion
Distilled water is the preferred and widely recommended choice for the humidifier bottles of oxygen machines. It minimizes mineral deposits, reduces the risk of microbial growth, protects delicate internal components, and contributes to patient comfort during long‑term oxygen therapy. Alternatives such as tap water, mineral water, or boiled water may appear convenient but can cause scaling, contamination, and reduced device life.
For medical facilities and pharmaceutical manufacturers, installing a reliable Distillation Water Machine and an integrated pure water system ensures that high‑quality distilled water is always available for oxygen machines and other sensitive applications. With the support of experienced suppliers like Everheal, these systems can be seamlessly built into modern, compliant, and efficient plant layouts.

FAQs About Distilled Water and Oxygen Machines
1. Is distilled water mandatory for every oxygen concentrator?
Distilled water is strongly recommended for almost all oxygen concentrators that use a humidifier bottle, because it prevents mineral buildup and helps maintain clean, consistent humidification. Users should always follow the specific instructions in their device's user manual, but “distilled water only” is one of the most common requirements.
2. How often should the distilled water be changed?
In most home and clinical situations, the distilled water in the humidifier bottle should be checked every day and completely replaced at least once daily. If the water becomes cloudy, discolored, or develops any odor, it should be discarded immediately, the bottle cleaned, and fresh distilled water added.
3. Can I use boiled tap water instead of distilled water?
Boiled tap water is not an ideal replacement for distilled water. While boiling may reduce some microorganisms, it does not remove minerals that cause limescale and deposits. Long‑term use of boiled tap water in an oxygen machine humidifier can lead to scaling, reduced performance, and potential damage to internal components.
4. What happens if tap water is used in the oxygen machine humidifier?
Using tap water can cause mineral deposits to form inside the humidifier bottle and associated tubing. Over time, these deposits may restrict flow, reduce the effectiveness of humidification, and put extra stress on the machine. Tap water can also support microbial growth if it is allowed to stand, increasing the risk of respiratory irritation or infection.
5. Why should hospitals and pharma plants install a Distillation Water Machine?
Hospitals and pharmaceutical facilities need large volumes of consistent, high‑purity water for oxygen humidifiers, sterilization, and drug manufacturing. A Distillation Water Machine provides a dependable, on‑site source of distilled water that can be integrated into a broader pure water and pure steam system. This setup simplifies quality control, ensures compliance with regulatory standards, and supports safe, reliable oxygen therapy.





















































