Direct-heating and indirect-heating clean steam generators both support reliable autoclave sterilization, but they differ significantly in steam source, control, lifecycle cost and regulatory risk for large-scale pharmaceutical plants. For a company like Ningbo Everheal Medical Equipment that designs full sterile utilities and autoclave lines, choosing the right concept is a strategic decision rather than a simple equipment purchase. [en.paulmueller]
Understanding Clean Steam for Autoclaves
Clean steam is steam generated from purified water in equipment specifically designed to avoid contamination from materials, additives, and boiler chemicals. In pharma and biopharma, regulators expect clean or pure steam whose condensate meets WFI-level or at least Purified Water specs for conductivity, TOC, and endotoxins. [linkedin]
For large autoclaves, saturated clean steam is preferred because:
- It delivers high latent heat transfer and fast, uniform sterilization. [electrosteam]
- A dryness fraction around 0.95 (95% dry) with minimal non‑condensable gases is considered the gold standard. [linkedin]
- Condensate quality at the point of use can be sampled and qualified as part of the steam system lifecycle. [gmpsop]
This quality focus is why the choice between direct- and indirect-heating generators matters so much in layout planning and validation strategies. [tlv]

What Do "Direct" and "Indirect" Heating Mean?
In pharmaceutical clean steam systems, the distinction is about how energy is transferred into the purified feedwater:
- Indirect-heating clean steam generators
- Use "house steam" or another heating medium inside a heat exchanger to boil purified water (RO/DI/WFI) on the secondary side. [electrosteam]
- No direct contact between heating steam and clean steam; energy crosses a tube wall or plate. [electrosteam]
- Direct-heating (direct steam injection or electric) clean steam generators
- Either inject heating steam directly into purified water (in some designs), or use direct electric immersion heaters in the clean-water vessel to generate steam. [watertreatment.en.made-in-china]
- The clean steam is produced in a dedicated stainless steel vessel with carefully controlled feed water and separation internals. [meco]
From a user's perspective as an engineering manager or validation lead, indirect systems feel like an extension of the boiler house, while direct systems feel like a standalone critical utility located close to the autoclaves.
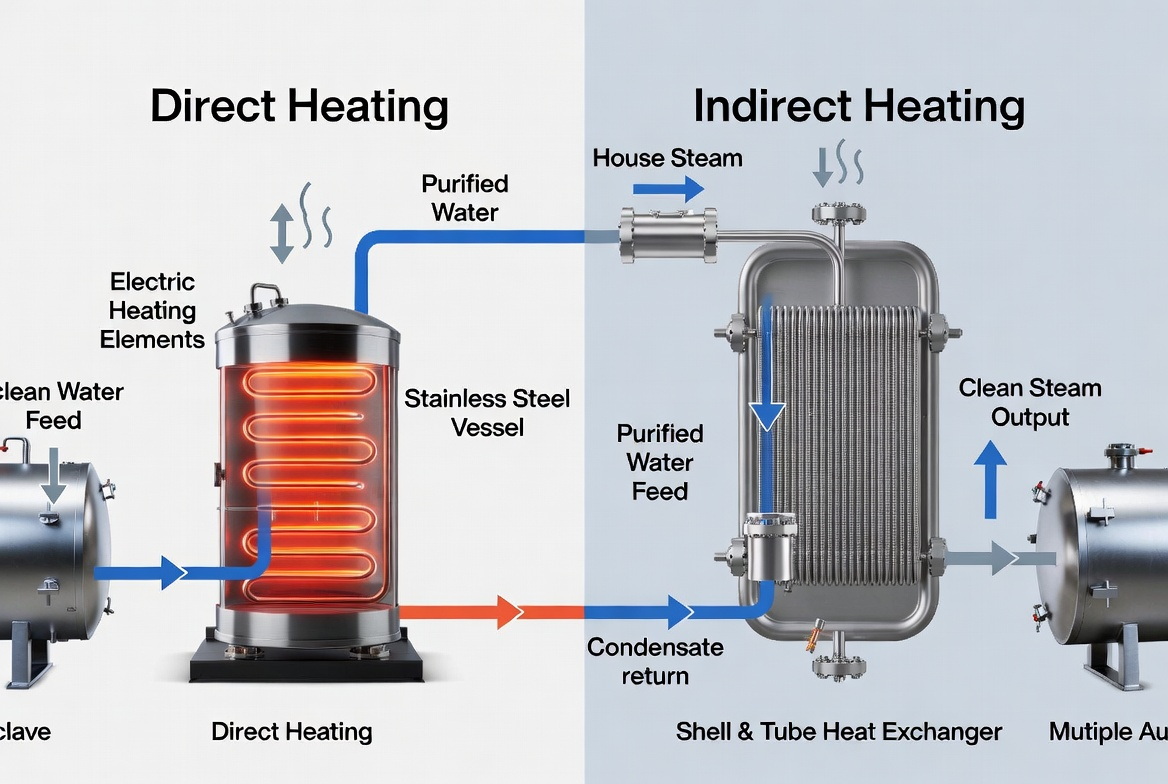
Direct- vs Indirect-Heating: Side‑by‑Side Comparison
Key technical and business differences
| Dimension | Direct-Heating Clean Steam Generator | Indirect-Heating Clean Steam Generator |
| Heating source | Direct steam injection or electric elements. (watertreatment.en.made-in-china) | Uses existing house steam on primary side of heat exchanger. (electrosteam) |
| Steam contact | May involve closer coupling to heating steam, or purely electrical heating of clean side. (watertreatment.en.made-in-china) | No direct contact; physical barrier between house steam and clean steam. (electrosteam) |
| Typical applications | Dedicated autoclaves, local SIP skids, where electrical power or standalone skid is preferred. (electrosteam) | Plants with large central boilers and widespread SIP/CIP networks using house steam energy. (electrosteam) |
| Installation footprint | Often compact skid; suited for point‑of‑use or near‑use installation. (meco) | Larger systems integrated into central utility areas or main mechanical rooms. (meco) |
| Control complexity | Typically simpler control loops focused on level, pressure, temperature and feed conductivity. (electrosteam) | Must coordinate house‑steam conditions, exchanger performance, and secondary steam control. (electrosteam) |
| Steam quality risk | Strongly dependent on vessel design, separation internals and feedwater quality. (linkedin) | Adds a barrier against boiler chemicals but inherits any instability from house steam parameters. (electrosteam) |
| Energy efficiency | Good local efficiency; electric models benefit from precise modulation but depend on electricity price and grid reliability. (electrosteam) | High system efficiency in sites with optimized central boilers and heat recovery. (electrosteam) |
| Scale | Well‑suited to single or a few large autoclaves and clustered users. (meco) | Ideal for multi‑line plants with many sterilization points and long distribution loops. (meco) |
| Validation & sampling | Easier to isolate as a single qualified utility feeding specific loads. (gmpsop) | Requires system‑level qualification (boiler → house steam → exchanger → clean steam header → autoclaves). (gmpsop) |
| Capex vs Opex | Lower capex for self‑contained skids; opex driven by electricity or high‑pressure steam consumption. (watertreatment.en.made-in-china) | Higher initial integration cost; lower marginal opex when leveraging existing boiler capacity. (electrosteam) |
Clean Steam Quality and Regulatory Expectations
Regulators and industry bodies such as ISPE focus on measurable steam attributes rather than generator type alone. For both direct- and indirect-heating, you must demonstrate that: [gmpsop]
- Condensate meets the relevant pharmacopeial limits for conductivity, TOC and endotoxin when required (Purified Water or WFI level). [robenmfg]
- Dryness fraction is typically at or above 0.95, with minimal superheat, to ensure efficient microbial kill in autoclaves. [linkedin]
- Non‑condensable gases remain below defined thresholds through design and routine sampling. [tlv]
A risk-based commissioning and qualification approach often targets:
1. Generator outlet (condensate sample) for baseline steam quality. [gmpsop]
2. Worst‑case points on the autoclave supply header, factoring in elevation, distance and trapping strategy. [ispeboston]
From my experience working with validation teams, distribution design (slope, trap placement, dead legs) creates more practical deviations than the choice between direct vs indirect heaters, but the heater concept sets the baseline for achievable quality. [electrosteam]
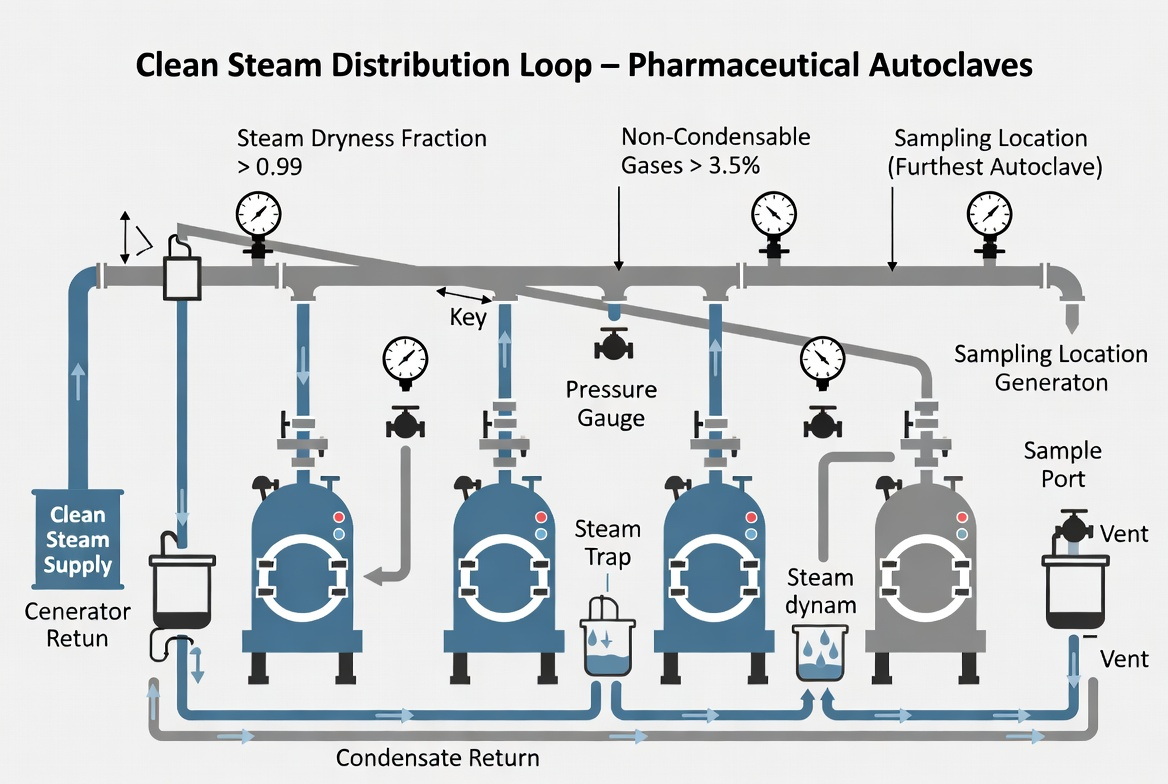
How Direct-Heating Clean Steam Generators Perform in Large Autoclave Lines
From a plant engineering standpoint, direct-heating clean steam generators are attractive when you need local, highly controllable steam right next to critical autoclaves.
Advantages
- Fast response and tight pressure control at the autoclave inlet, which supports robust sterilization cycles and shorter recovery times between loads. [electrosteam]
- Compact skid design makes them easy to position near sterilizers, minimizing distribution losses and non‑condensable gas accumulation. [meco]
- Electric or dedicated steam heating avoids exposing the clean steam side to boiler treatment chemicals, provided the design maintains separation where needed. [watertreatment.en.made-in-china]
For Chinese manufacturers exporting to regulated markets, this concept often simplifies modular line export: you can ship an integrated autoclave + clean steam skid that the end user connects to electrical power and a purified water loop, rather than relying on local boiler infrastructure. [en.paulmueller]
Limitations
- In plants with many autoclaves and SIP users, operating several independent direct-heating units can raise energy costs and complicate maintenance scheduling. [meco]
- If direct steam injection from house steam is used, you must carefully control and document chemical treatment programs and particulate filters to maintain acceptable condensate quality. [tlv]
Expert insight: In projects where Ningbo Everheal‑type suppliers deliver turnkey sterile lines, we often treat direct-heating pure steam generators as "autoclave companions"—they live in the same layout, share a control strategy, and are qualified as one integrated system.
How Indirect-Heating Clean Steam Generators Perform in Large Facilities
Indirect clean steam generators are typically the preferred option for high‑throughput, campus‑scale pharmaceutical facilities. [electrosteam]
Advantages
- They reuse existing house steam from central boilers as the energy source, which is cost‑effective when the boiler plant is already optimized. [electrosteam]
- The heat exchanger barrier keeps boiler treatment chemicals and corrosion products out of the clean steam circuit, reducing contamination risk. [electrosteam]
- It is easier to scale up to multiple tons per hour of pure or clean steam, feeding many autoclaves, SIP points and humidifiers from the same header. [meco]
In practice, indirect systems become the backbone of a site‑wide clean utility, with autoclaves treated as just one of several critical users.
Limitations
- Autoclave performance becomes sensitive to house steam stability; pressure or quality fluctuations on the primary side can impact secondary steam conditions. [electrosteam]
- Distribution systems must be carefully designed (slope, traps, minimal dead legs) to maintain steam quality all the way to the most distant sterilizer. [ispeboston]
- Centralization can make incremental expansions slower and heavier on capex when you add new buildings or sterilizers in remote areas of the site. [electrosteam]
Design Considerations for Large-Scale Autoclave Sterilization
When Ningbo Everheal or similar integrators design a full line, the selection between direct and indirect should be embedded in a structured user requirement and risk analysis rather than handled as a catalog choice.
Key design parameters to evaluate:
- Sterilization load profile: Number of autoclaves, chamber sizes, turnaround time, peak vs average load. [electrosteam]
- Steam quality targets: Whether condensate must consistently meet WFI specs or Purified Water levels, and the acceptable limits for non‑condensables. [robenmfg]
- Existing utilities: Availability and stability of house steam, purified water loops, and electrical power. [electrosteam]
- Regulatory environment: Expectations from FDA, EMA, NMPA or local authorities regarding sampling, documentation and periodic requalification. [linkedin]
A practical method I use with project teams is to build a simple decision matrix that scores direct vs indirect across risk, capex, opex and flexibility for each site. In many cases, the output suggests a hybrid architecture: indirect central generation for baseline demand, supplemented by local direct-heating pure steam modules near expansion autoclaves.
Step‑by‑Step Selection Framework for Engineering Teams
To turn these principles into actionable steps, engineering and QA can follow a structured path:
1. Define autoclave demand and criticality
Map all current and planned autoclaves, their chamber sizes, load types, and sterilization cycles to estimate peak and average clean steam demand. [electrosteam]
2. Assess current utility infrastructure
Evaluate boiler capacity, house steam pressure stability, purified water generation and storage, and electrical redundancy. [electrosteam]
3. Set explicit steam quality requirements
Agree on condensate conductivity, TOC, endotoxin limits, dryness targets and sampling points with QA and validation. [robenmfg]
4. Score direct vs indirect options
Compare both concepts against risk, capex, opex, maintainability, and layout constraints using a weighted scorecard. [tlv]
5. Develop a distribution concept
Design headers, branch lines, traps, and sample ports to maintain steam quality across autoclave locations. [electrosteam]
6. Plan commissioning and qualification
Define FAT/SAT, IOQ testing, steam quality sampling strategy and periodic requalification aligned with relevant GMP guidance. [gmpsop]
As a cleanroom and equipment supplier, Ningbo Everheal can build this methodology into its front‑end engineering design (FEED) service, positioning the company not just as an equipment maker but as a long‑term process utility partner.
Where Ningbo Everheal's Pure Steam Solutions Fit In
Manufacturers of pure steam generators designed to meet USP/EP/JP/CP standards are already providing:
- Stainless steel generator vessels with sanitary construction (e.g., sloped piping, double tube sheets, minimal dead legs). [meco]
- Capability to produce steam that condenses to Purified Water or WFI specifications, supporting critical autoclave and SIP uses. [en.paulmueller]
- Flexible capacity ranges from a few hundred to tens of thousands of pounds per hour. [meco]
For a company like Ningbo Everheal, this capability can be framed as:
- Direct-heating pure steam modules integrated near autoclaves in modular lines aimed at CDMOs, biologics pilot sites and hospital sterile centers.
- Indirect-heating clean steam generators tied into house steam systems in large vaccine, oncology, and injectable plants with centralized utility architecture.

Practical Recommendation Summary for Buyers
For procurement, engineering and QA managers comparing options:
- Choose direct-heating clean steam generators when you need localized, tightly controlled steam dedicated to a cluster of autoclaves, especially in modular or greenfield projects with limited house steam infrastructure. [electrosteam]
- Choose indirect-heating clean steam generators when you already operate a robust boiler plant and want to supply a large autoclave population and SIP network from a central clean steam header. [electrosteam]
- In many complex sites, a hybrid strategy combining both concepts offers the best balance between reliability, cost and expansion flexibility. [tlv]
FAQs
1. Is clean steam always required for autoclave sterilization in pharma?
Clean or pure steam is strongly preferred in regulated pharmaceutical environments because it avoids boiler additives and contaminants; condensate can be verified against Purified Water or WFI limits. Some non‑critical utilities may still use plant steam, but critical product-contact items and load types typically require validated clean steam. [electrosteam]
2. Does indirect heating guarantee better steam quality than direct heating?
Not automatically. Steam quality ultimately depends on feed water, generator design, control and distribution rather than heating concept alone. Indirect systems do offer a physical barrier to boiler chemicals, but poor house steam or bad distribution can still compromise condensate quality. [linkedin]
3. How often should steam quality be tested for large autoclave systems?
A risk‑based program typically includes testing during commissioning and qualification, at defined intervals (for example annually or semi‑annually) and after significant changes to the system. Critical parameters such as conductivity, dryness and non‑condensable gases are usually checked at both the generator outlet and representative points of use. [gmpsop]
4. Can one clean steam generator feed both autoclaves and SIP/CIP systems?
Yes. Clean and pure steam generators are commonly sized and designed to serve autoclaves, SIP, CIP and sometimes humidification, provided distribution is correctly engineered and demand diversity is understood. However, autoclaves with very tight cycle requirements may still justify dedicated generators, especially in high‑risk manufacturing areas. [electrosteam]
5. What are typical failure modes that affect autoclave performance?
Frequent issues include wet steam from poor drainage, non‑condensable gas pockets due to bad venting, and conductivity excursions from contaminated feed water or internal corrosion. These problems manifest as failed biological indicators, extended cycles, or repeated deviations in sterilization records. [ispeboston]
Call to Action
If you are planning or upgrading a large‑scale autoclave sterilization line, the choice between direct and indirect clean steam generators should be validated early in your layout and utility design. Ningbo Everheal Medical Equipment can support you with concept selection, capacity sizing, 3D layout planning and integrated pure steam generator–autoclave solutions aligned with current GMP expectations.
References
1. Electro-Steam, "Implementing Steam Sterilization in Pharmaceutical Facilities." [electrosteam]
2. Electro-Steam, "Indirect Clean Steam Generator." [electrosteam]
3. Electro-Steam, "Clean Steam Generators for Pharma." [electrosteam]
4. MECO, "Pure Steam & Clean Steam Generators – Biopharmaceutical Applications." [meco]
5. Roben Manufacturing, "Clean Steam Generation for Pharmaceutical Manufacturing." [robenmfg]
6. TLV, "Clean & Pure Steam – Steam Basics and Quality Factors." [tlv]
7. ISPE Boston, "Water and Steam – Pharmaceutical Steam Sampling." [ispeboston]
8. GMP SOP Sample, "Clean Pure Steam System Commissioning and Qualification – Sampling Strategy." [gmpsop]
9. LinkedIn, S. Yeotikar, "Steam Quality: The Hidden Variable in Steam Sterilization." [linkedin]
10. Paul Mueller Company, "Pure Steam Generator – Pharmaceutical." [en.paulmueller]
11. Made-in-China, "Electric Heating Clean Steam Generator – Sterilization Electric Pure Steam Generator." [watertreatment.en.made-in-china]
12. Electro-Steam, "Indirect Clean Steam Generators in Biopharma and Pharma." [electrosteam]
13. Electro-Steam, "Clean Steam System Design Fundamentals." [electrosteam]






















































