As a process engineer supporting global GMP projects in China and overseas, I have seen both single RO and two‑stage RO + EDI succeed—or fail—on high‑silica feedwaters, depending on design and operation. In this article, I will compare these two routes from a real project and industry‑expert perspective, focusing on pharmaceutical purified water preparation lines like those delivered by Ningbo Everheal Medical Equipment Co., LTD. [pharmaguideline]
High‑silica raw water is especially challenging in pharma because silica behaves differently from hardness salts: it is only weakly ionized, often passes through standard softening, and tends to form stubborn scale on membranes and heat‑transfer surfaces. The wrong choice at the design stage can lock a plant into chronic fouling, frequent CIP, and unstable conductivity. The right choice gives a decade or more of stable compliance with USP/EP water specs and predictable OPEX. [ske-eagle]
Understanding High-Silica Feedwater in Pharma Projects
From a water‑system designer's viewpoint, "high‑silica" is less about an absolute number and more about risk of scaling and margin to design limits. [aqualitek]
Typical pain points with high‑silica raw water in pharma utilities include: [molewater]
- Silica in raw water often above 20–30 mg/L as SiO₂, especially in surface and well waters.
- Silica combines with calcium and magnesium to form low‑solubility silicate scales on RO membranes.
- Silica passes through softeners; classic hardness‑focused design underestimates its impact.
- Hot PW loops and multi‑effect distillation units are prone to long‑term silica deposition if pretreatment is weak.
For GMP plants, the consequences are not only higher chemical and maintenance costs, but also qualification risks: unstable conductivity, frequent shutdowns for membrane cleaning, and excursions in TOC or microbiological performance. [pharmaguideline]

Option 1 – Reverse Osmosis-Only Systems
How RO-Only Systems Work in Pharma
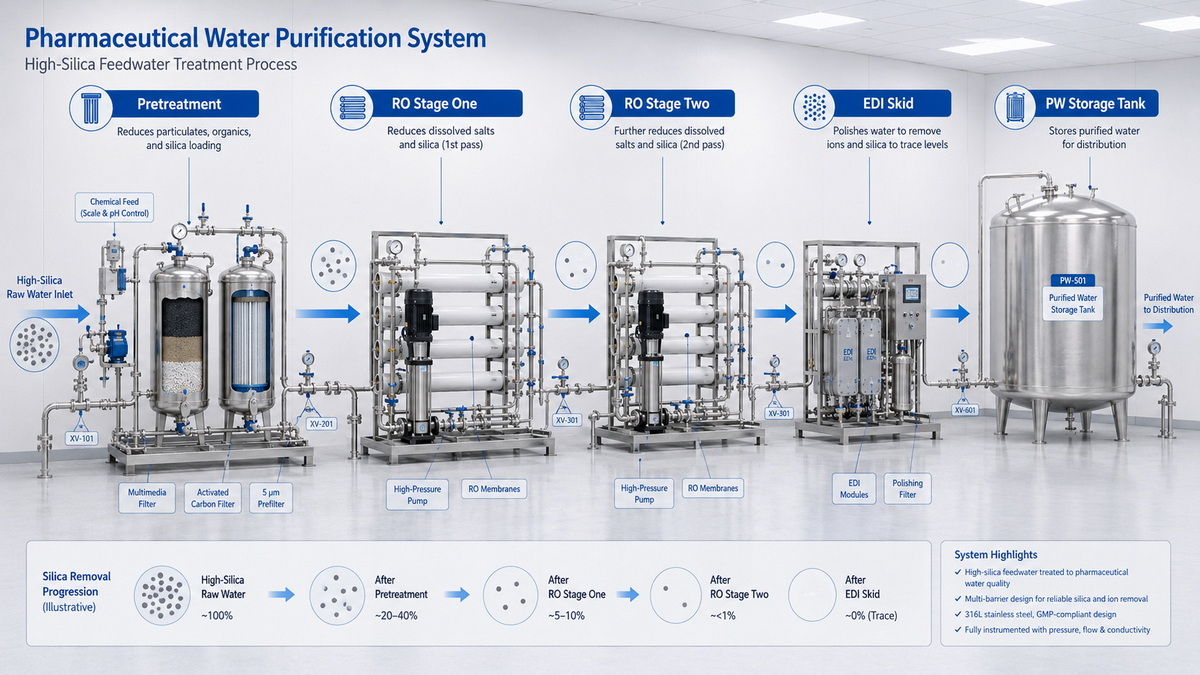
A typical single‑stage RO purified water system in a pharmaceutical facility consists of: [pharmaguideline]
- Pre‑treatment: multi‑media filter, activated carbon, softener, micron filters, chemical dosing.
- Single RO pass: high‑pressure pump feeding spiral‑wound RO membranes.
- Post‑treatment: UV, ultrafiltration, polishing filters, storage tank and distribution loop.
RO membranes remove over 98% of dissolved ions, organics, particles, and microorganisms under correctly designed conditions, making RO the workhorse of modern pharma water generation. For moderate‑silica waters with proper antiscalant and recovery control, single RO can provide purified water that meets pharmacopoeial specifications. [ske-eagle]
Advantages of RO-Only for Pharma Prep
From a UX and OPEX perspective, RO‑only systems offer clear benefits: [webofpharma]
- Lower capital cost: Fewer unit operations, no EDI modules or high‑voltage DC supplies.
- Simpler operation: Operators already understand RO; automation is straightforward.
- Lower energy per unit than multi‑effect distillation, especially for PW/HPW grades.
- Smaller footprint and simpler skid integration.
For mid‑size generic plants where raw water is relatively clean and silica is moderate, RO‑only remains a cost‑effective choice for PW. [pharmaguideline]
Limitations of RO-Only on High-Silica Feedwater
However, on high‑silica and variable feedwaters, RO‑only quickly hits its limits: [aqualitek]
- Silica scaling: As recovery increases, silica concentration in the concentrate rises, surpassing solubility and depositing on membranes.
- Tight recovery limits: To keep silica below scaling thresholds, recovery must be kept low, increasing feedwater consumption and wastewater volume.
- Frequent chemical cleaning: High‑silica plants often require more CIP cycles, shortening membrane life.
- Polishing gaps: Meeting stricter conductivity specs or future upgrades (e.g., to HPW) can be difficult without an additional polishing step.
In practice, many "RO‑only" designs dealing with high‑silica feedwaters silently turn into RO + intensive antiscalant + frequent CIP designs, absorbing OPEX and reliability risks over time. [molewater]
Option 2 – Two-Stage RO + EDI Systems
Core Principle of RO + EDI
In a two‑stage RO + EDI system, RO and EDI play complementary roles: [ske-eagle]
- First RO stage: removes the majority of ions, organics, and microbes, creating low‑load permeate.
- Second RO (polishing RO): further reduces TDS, hardness, and silica, particularly under optimized recovery control.
- EDI unit: uses ion‑exchange resins plus a DC electric field to continuously remove residual ions without chemical regeneration, pushing water quality into ultrapure range.
Industry data shows that EDI is typically fed with RO permeate at <20–50 µS/cm conductivity and hardness <0.1–0.2 mg/L as CaCO₃. On high‑silica waters, dual RO staging is often used precisely to achieve this stable feed quality. [ske-eagle]
Advantages of Two-Stage RO + EDI for High-Silica
For high‑silica pharmaceutical projects, a two‑stage RO + EDI line delivers several critical advantages: [webofpharma]
- Superior ion removal: EDI "polishes" RO permeate to reach very low conductivity and silica levels suitable for high‑end PW and HPW.
- Continuous deionization: No batch resin regeneration, no acid/alkali handling, minimal chemical storage.
- Better long‑term stability: EDI resins are continuously regenerated by the electric field, so performance remains stable over long campaigns.
- Scalability: Easy to scale up for biotech and injectable drug lines needing tighter water specs.
- Lower lifecycle cost: Higher CAPEX but lower chemical, labor, and compliance risk over the equipment's lifecycle.
For Everheal's target customers—global formulators, injectables plants, and biotech facilities—this route aligns closely with modern GMP and sustainability expectations. [everhealgroup]
Challenges and Design Sensitivities with RO + EDI
At the same time, RO + EDI is less forgiving of weak design: [webofpharma]
- Higher initial investment: EDI modules, DC power, and control hardware add upfront cost.
- Strict feedwater requirements: Residual hardness, chlorine, CO₂, and organics must be tightly controlled, or fouling occurs.
- Need for expert design and O&M: Module sizing, current density, and cleaning strategies require specialized know‑how.
This is exactly where a specialized OEM like Ningbo Everheal adds value: configuring pre‑treatment, dual RO, and EDI layout for each site's silica profile and regulatory target. [everhealgroup]
Expert Comparison – RO Vs. Two-Stage RO + EDI for High-Silica Feedwater
Key Technical and UX Factors Side by Side
Below is a practical comparison from a pharma‑project perspective: [aqualitek]
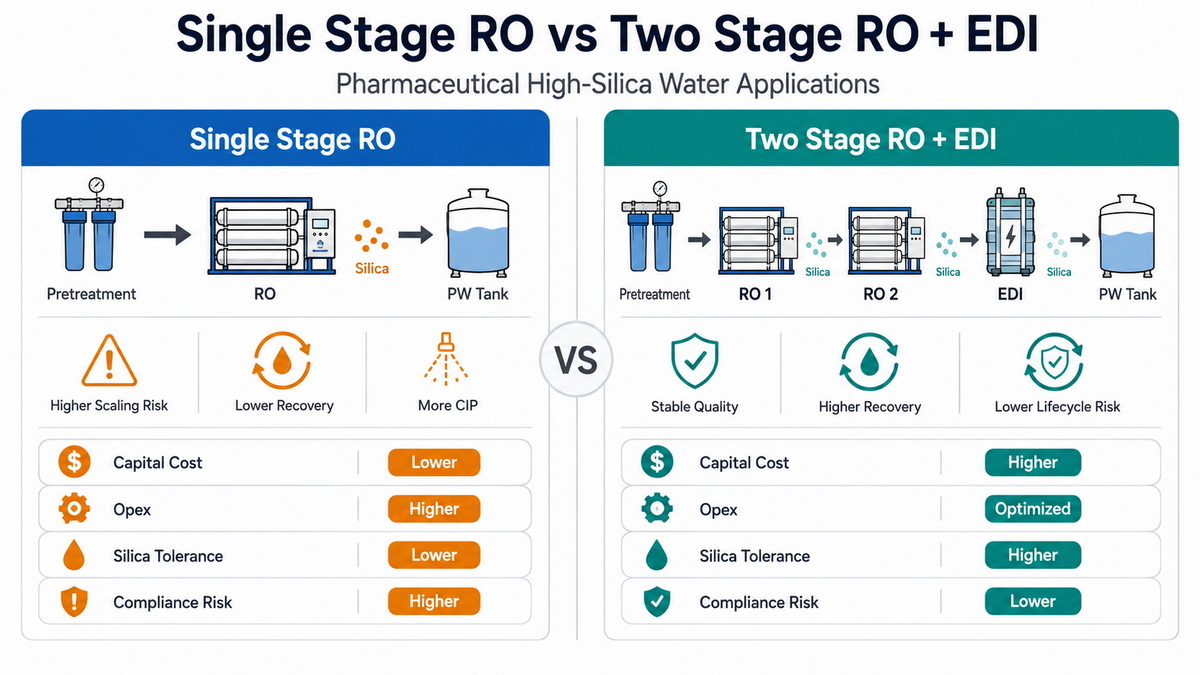
| Factor | Single-Stage RO System | Two-Stage RO + EDI System |
| Target water grade | PW for solid or non‑critical liquids | PW, HPW; easy upgrade to tighter specs |
| High‑silica tolerance | Moderate (requires low recovery, strong antiscalant) | High (dual RO controls silica, EDI polishes residual) |
| Typical recovery | Lower on high‑silica (to avoid scaling) | Higher overall with smart staging |
| Water quality stability | Good, but sensitive to feed swings | Very stable, EDI buffers minor feed variations |
| Chemical consumption | Antiscalant, CIP chemicals | Minimal regeneration chemicals, mainly CIP/cleaning |
| Capital cost | Lower | Higher initial CAPEX |
| Lifecycle OPEX | Higher on difficult waters | Lower when used for high‑spec, long‑term plants |
| Compliance risk | Higher for future spec tightening | Lower, easier to meet stricter future standards |
| Operator skill requirement | Moderate | Higher, but automation reduces daily workload |
From a user‑experience angle, dual RO + EDI typically delivers a more predictable, "set‑and‑forget" purified water quality for high‑silica sites, once the system is properly commissioned and validated. [ske-eagle]
Realistic Pharma Scenario – When High-Silica Breaks RO-Only Design
Consider a mid‑size injectable plant in Asia sourcing raw water from a coastal groundwater source with silica fluctuating between 25–45 mg/L and seasonal TDS changes. [molewater]
With an RO‑only design, we often see these patterns: [ske-eagle]
1. Initial operation at "optimistic" recovery.
2. Silica scaling appears within months, evidenced by rising differential pressure and falling permeate flow.
3. Operators respond with frequent CIPs, aggressive antiscalant dosing, and recovery reduction.
4. Water cost and downtime rise; qualification events become stressful because the system has no polishing margin.
In contrast, the same site engineered with two‑stage RO + EDI can: [aqualitek]
- Run the first RO at conservative recovery focused on bulk removal and silica control.
- Use the second RO and carbonate control to deliver stable, low‑TDS, low‑silica permeate to EDI.
- Let EDI handle the final polishing, absorbing small feed swings without affecting loop quality.
In practical terms, QA sees fewer excursions, maintenance has predictable tasks, and production has a stable purified water backbone for batch scheduling. [molewater]
Design Checklist for High-Silica Pharma Water Systems
Step-by-Step Approach Used by Industry Experts
When advising clients, I use a simple 5‑step framework for high‑silica sites:
1. Characterize feedwater properly
Include seasonal silica, hardness, TDS, organic load, and microbiology; collect data over time, not just a single sample. [ske-eagle]
2. Define regulatory and business targets
Decide whether you only need PW today, or plan to upgrade to HPW/biologics in 3–5 years; align with USP, EP, and local GMP expectations. [pharmaguideline]
3. Select core technology route
- Moderate silica, simple oral solid plant → RO‑only can be acceptable.
- High silica, injectables, biotech, export‑oriented GMP → two‑stage RO + EDI is usually the safer choice. [ske-eagle]
4. Engineer pretreatment for silica risk
Leverage custom softening, antiscalant selection, and recovery control to keep silica below scaling thresholds; focus on stable feed to RO and EDI. [aqualitek]
5. Plan lifecycle O&M from day one
Build CIP strategies, membrane/EDI module replacement, online monitoring, and spare‑parts management into the URS and design. [ske-eagle]
A partner like Ningbo Everheal can integrate these steps into a turnkey solution—from purified water preparation skids to pure steam, distillation, and filling line utilities—so the water system is fully aligned with plant layout and production strategy. [everhealgroup]
Expert Insights on EDI in Modern Pharma Plants
Why EDI Is Becoming the Default Polishing Technology
Globally, EDI combined with RO is increasingly recognized as a modern, sustainable choice for pharmaceutical purified water systems: [webofpharma]
- No chemical regeneration: Avoids handling and discharge of strong acids and bases used in traditional mixed‑bed DI.
- Lower environmental impact: Supports ESG and corporate sustainability goals.
- High automation potential: Integrates well with PLC/SCADA and data integrity requirements.
- Regulatory alignment: Widely accepted for PW and HPW under USP/EP when properly qualified.
Industry guides emphasize that, for many new facilities, RO + EDI is now the reference design rather than an upgrade option. [molewater]
Typical Specification Targets Upstream of EDI
To protect the EDI module and ensure stable performance, most vendors require: [pharmaguideline]
- RO permeate conductivity below 20–50 µS/cm.
- Hardness <0.1–0.2 mg/L as CaCO₃.
- Low residual chlorine and oxidants.
- Controlled CO₂ and silica through pretreatment and RO staging.
In high‑silica projects, dual‑stage RO is often used not only for conductivity but specifically to keep silica and hardness within EDI design windows. [ske-eagle]
Where Everheal Fits in Your High-Silica Water Strategy
As a Chinese manufacturer of purified water preparation systems, pure steam generators, and multi‑effect distillation units, Ningbo Everheal Medical Equipment Co., LTD specializes in end‑to‑end utilities for pharmaceutical plants. [everhealgroup]
For customers dealing with high‑silica feedwaters, Everheal can: [everhealgroup]
- Analyze your feedwater and production plan to recommend RO‑only versus two‑stage RO + EDI.
- Design custom pre‑treatment, including softeners, antiscalant dosing, and filtration tailored to silica risk.
- Supply integrated RO, EDI, pure steam, and MWS systems aligned with your filling, sterilization, and plant layout.
- Support DQ/IQ/OQ/PQ and provide documentation compatible with global inspections.
This integration means the purified water system is not an isolated skid, but part of a coherent, GMP‑oriented plant concept.

Final Recommendation – Which System Is Best for High-Silica Pharma Prep?
From both a technical and business standpoint, my recommendation is:
- For moderate silica and basic PW needs, a well‑engineered RO‑only system can be justified if lifecycle risks are explicitly accepted.
- For truly high‑silica feedwater, sites planning injectable or export‑oriented production, or plants expecting tighter future specifications, a two‑stage RO + EDI system is usually the best long‑term choice. [aqualitek]
Combined with professional pretreatment design and lifecycle support, dual RO + EDI provides a robust, GMP‑ready backbone for modern pharma facilities.
Call to Action – Plan Your High-Silica Water System with Everheal
If your upcoming or existing plant faces high‑silica feedwater and stringent GMP expectations, the most effective next step is a technical discussion based on your actual data.
You can share your raw water analysis and target water grades with Ningbo Everheal Medical Equipment Co., LTD, and request a tailored comparison of RO‑only versus two‑stage RO + EDI for your line capacity, layout, and budget. This allows you to convert a complex design decision into a clear, documented technical and financial choice. [everhealgroup]
FAQ: Reverse Osmosis Vs. Two-Stage RO + EDI in Pharma
Q1. Is RO alone enough for USP purified water on high-silica feedwaters?
In some cases yes, but only at conservative recovery and with strong antiscalant and cleaning strategies; in many high‑silica sites, lifecycle risks make two‑stage RO + EDI more robust. [molewater]
Q2. Does EDI remove silica completely?
EDI mainly removes charged ions; silica removal depends heavily on upstream pretreatment and RO staging, so EDI must be part of a broader silica‑aware design rather than relied upon alone. [ske-eagle]
Q3. How does RO + EDI affect validation and regulatory inspections?
Regulators accept RO + EDI as a modern, sustainable solution for PW/HPW when the system is properly designed, qualified, and monitored with appropriate online and offline controls. [pharmaguideline]
Q4. What are typical maintenance tasks for RO + EDI systems?
Tasks include routine sanitization, module integrity checks, CIP for RO and EDI when needed, filter replacement, and continuous monitoring of key parameters such as conductivity and TOC. [ske-eagle]
Q5. Can an existing RO-only plant be upgraded to RO + EDI?
In many cases, yes: a second RO stage and EDI skid can be added if space, piping, and electrical capacity allow; design review and re‑qualification are essential. [aqualitek]
References
1. SKE Eagle – "EDI Systems in Reverse Osmosis Purified Water: A Technical Analysis and Mainstream Brand Guide." [ske-eagle]
2. Pharmaguideline – "Types of Purified Water Systems in Pharmaceuticals." [pharmaguideline]
3. Ningbo Everheal Medical Equipment Co., LTD – Official Website and Company Profile. [everhealgroup]
4. Web of Pharma – "RO vs. EDI Water System." [webofpharma]
5. SKE Eagle – "Pharmaceutical Water System: A Comprehensive Guide to Modern Purification Solutions." [ske-eagle]
6. Aqualitek – "Choosing Electrodeionization Systems: Compare Key Specs." [aqualitek]
7. Molewater – "Top 5 Considerations When Selecting a Pharmaceutical Water Purification System." [molewater]






















































