WFI generation for energy‑constrained facilities is increasingly a strategic decision between multi‑effect distillation (MED) systems and newer membrane‑based ambient WFI solutions, especially for plants that must balance GMP compliance, OPEX, and sustainability. As a turnkey pharmaceutical utilities provider, Ningbo Everheal Medical Equipment Co., LTD can help clients design hybrid or phased layouts that keep regulatory confidence while progressively lowering energy intensity. [wateronline]
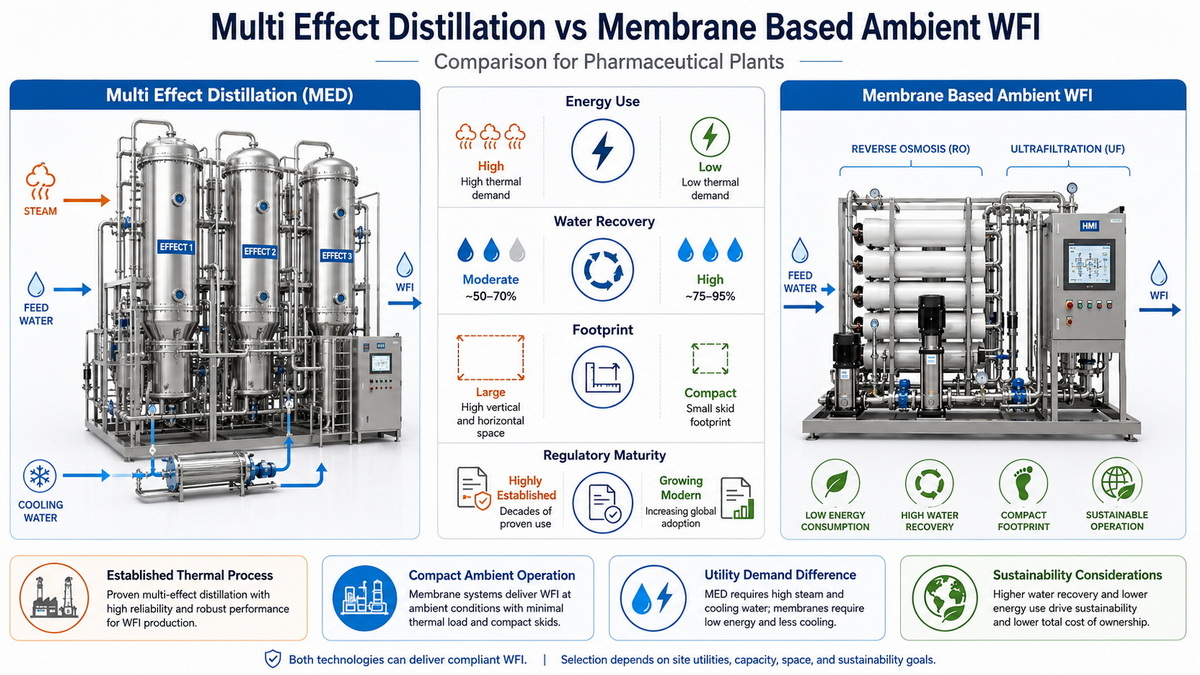
Understanding WFI Generation in Energy‑Constrained Facilities
Water for Injection (WFI) is one of the most energy‑intensive utilities in a pharma facility, often accounting for a large share of steam and cooling demand. Studies on large plants show that WFI production alone can consume a significant portion of a site's heating and cooling capacity, making the choice of generation technology critical when utilities are limited or expensive. [crbgroup]
In energy‑constrained or emerging markets, engineers must weigh the robustness and familiarity of multi‑effect distillation against the lower utility demand and reduced footprint of membrane‑based ambient systems. This decision is no longer purely technical; it is directly tied to carbon reduction goals, long‑term OPEX, and the feasibility of future capacity expansions. [processandcontrolmag.co]
What Is Multi‑Effect Distillation for WFI?
Multi‑effect distillation uses a series of evaporator "effects" where steam from one stage becomes the heating medium for the next, improving thermal efficiency compared with single‑effect stills. The process relies on high‑temperature evaporation and condensation, which inherently delivers hot, self‑sanitizing WFI suitable for direct feed to storage and distribution loops. [cy-water]
Because the process reaches high temperatures, MED systems provide intrinsic microbial control and strong endotoxin removal, which is why pharmacopeias have historically referenced distillation as the default approach for WFI. For global customers, this long regulatory track record makes multi‑effect distillation a low‑risk choice for greenfield sterile facilities and for markets where authorities still prefer distillation‑based WFI. [crbgroup]
What Are Membrane‑Based Ambient WFI Systems?
Membrane‑based ambient WFI systems typically combine high‑rejection reverse osmosis, advanced pretreatment, and ultrafiltration or other polishing steps to meet WFI conductivity, TOC, and microbial limits without bulk-phase evaporation. Unlike thermally driven stills, these systems operate near ambient temperature, using heat only for sanitization cycles or downstream distribution needs. [wateronline]
Recent pharmacopeial changes in regions such as Europe and the U.S. have explicitly allowed membrane‑based routes to WFI, provided that risk management, control strategies, and validation demonstrate equivalent quality and safety. This regulatory shift, combined with global energy pressure, has triggered a wave of interest in ambient WFI solutions, especially for new biotech and vaccine facilities aiming for sustainability certifications. [csrwire]
Side‑by‑Side Comparison: MED Vs. Membrane‑Based Ambient WFI
The table below summarizes key decision criteria for energy‑constrained facilities.
WFI Generation Options in Energy‑Constrained Plants
| Dimension | Multi‑Effect Distillation (MED) | Membrane‑Based Ambient WFI |
| Core principle | Thermal evaporation in multiple stages using cascading steam energy (crbgroup) | Multi‑stage membrane filtration (RO, UF, polishing) at ambient temperature (wateronline) |
| Energy profile | High steam demand, significant cooling and blowdown; moderate electricity (crbgroup) | Lower overall energy; significantly reduced steam and cooling, higher but manageable electricity (wateronline) |
| Water recovery | More waste from blowdown and higher pretreatment rejection (crbgroup) | Better water utilization due to lower pretreatment rejection (crbgroup) |
| Temperature & sanitization | Delivers hot, self‑sanitizing WFI; strong microbial robustness (crbgroup) | Ambient WFI; relies on chemical/thermal sanitization cycles and tight monitoring (wateronline) |
| Regulatory perception | Long‑standing global acceptance, very familiar to regulators (crbgroup) | Increasingly accepted; needs more detailed risk assessments and lifecycle validation (crbgroup) |
| Footprint | Larger skids, associated steam and cooling utilities, and equipment rooms (crbgroup) | Smaller footprint, simpler pipework and reduced utility rooms (wateronline) |
| CAPEX | Higher initial investment for stills, boilers, and cooling capacity (crbgroup) | CAPEX can be lower overall, especially where steam and cooling capacity would need upgrades (wateronline) |
| OPEX | Sensitive to fuel or steam price volatility, higher maintenance on steam side (crbgroup) | Lower utilities cost over lifecycle, but requires membrane replacement and higher instrumentation density (wateronline) |
| Best suited for | Plants prioritizing robustness, hot WFI, and conservative regulatory expectations | Energy‑constrained or sustainability‑driven plants with strong QA/QC culture |
[cy-water]
From an engineer's perspective, MED remains the conservative "workhorse," while membrane‑based ambient systems are the strategic choice for long‑term energy and water savings. [csrwire]
Energy and Sustainability Considerations
Independent assessments of large pharma sites show that WFI production by distillation can dominate natural gas (steam) usage and impose heavy cooling loads. By contrast, membrane‑based ambient systems can dramatically reduce steam consumption—one study reported almost one‑third lower steam use compared with an existing multi‑effect still—while also cutting water waste. [crbgroup]
For energy‑constrained facilities, this difference is critical:
- Reduced steam demand frees boiler capacity for process and HVAC, or allows smaller, more efficient boilers. [austar.com]
- Lower cooling needs ease the load on chillers and cooling towers, which is important where ambient temperatures are high or water availability is limited. [austar.com]
- Better water recovery supports corporate sustainability agendas by lowering intake and wastewater volumes. [crbgroup]
However, MED can still fit well when integrated with combined heat and power (CHP) or advanced building energy‑saving technologies, where recovered waste heat can support still operation without proportionally higher fuel consumption. In such layouts, the distillation system becomes part of a wider energy‑optimization strategy rather than a stand‑alone energy sink. [processandcontrolmag.co]
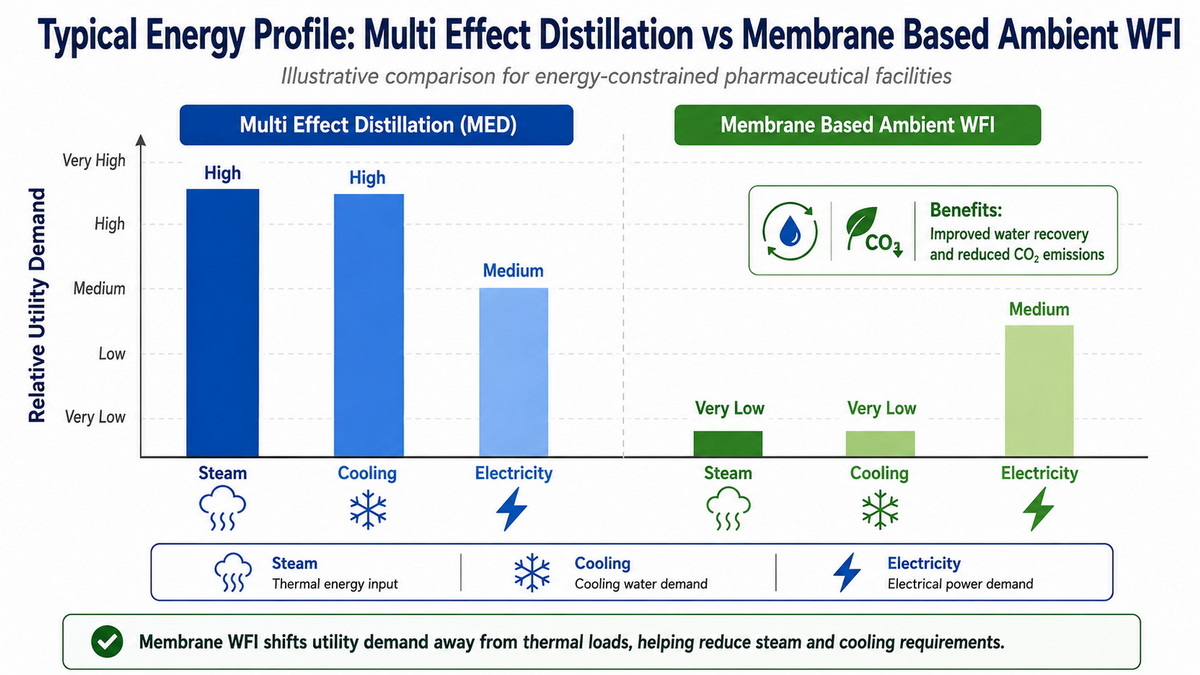
Risk, Reliability, and Regulatory Confidence
From a risk‑management standpoint, multi‑effect distillation offers inherent thermal barriers that simplify control strategies: high operating temperatures, low bioburden potential, and well‑understood failure modes. Operators and regulators have decades of experience interpreting alarm histories, deviation reports, and maintenance records for MED units. [cy-water]
Membrane‑based ambient systems, by design, operate in a microbiologically friendlier temperature range and rely on:
- Tight control of pretreatment and biofilm risk.
- Frequent sanitization and integrity testing of key barriers.
- Sophisticated monitoring for TOC, conductivity, and microbial counts.
[wateronline]
Regulators increasingly accept such systems, but they expect robust lifecycle documentation, including risk assessments, design qualification, and ongoing performance trending. For facilities with limited QA/QC resources, MED may therefore remain the safer choice, while larger or more mature organizations can comfortably exploit the efficiency of ambient WFI with rigorous automation and data analytics. [csrwire]
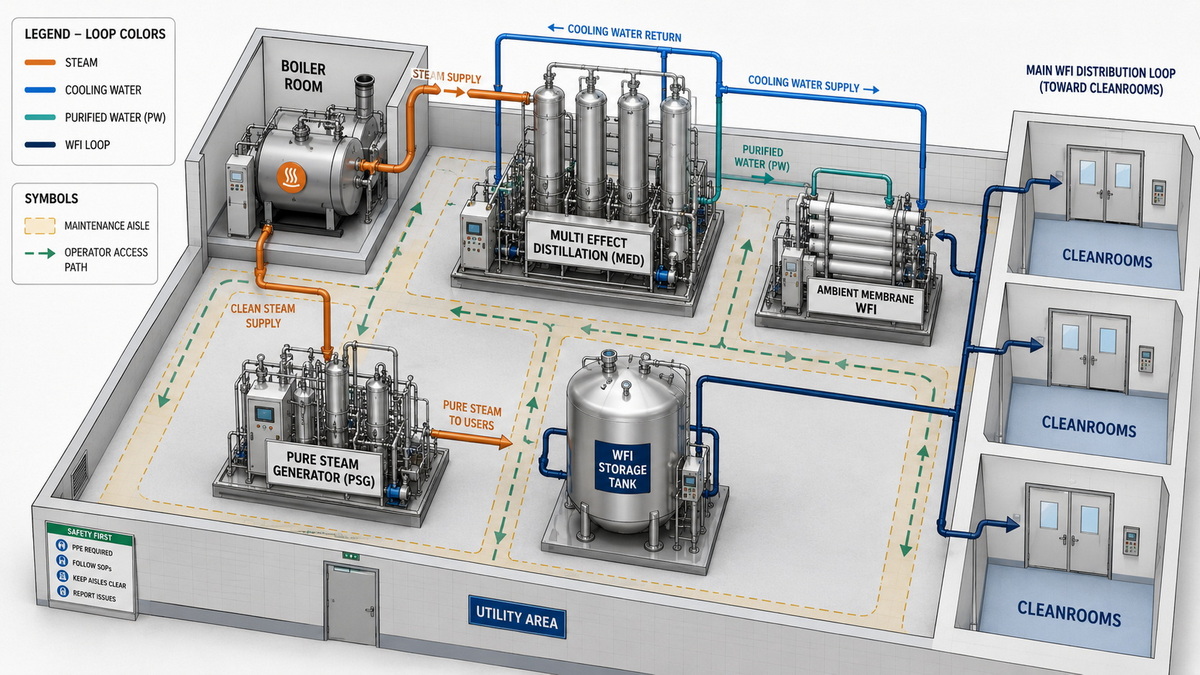
Layout and Footprint: How Everheal Designs for Real Sites
Ningbo Everheal Medical Equipment Co., LTD specializes in integrating purified water, pure steam, and distillation systems into GMP plant layouts, with an emphasis on compact, maintainable utility rooms and clear operator access. For multi‑effect distillation, Everheal's designs often cluster stills, pure steam generators, and WFI storage close to boiler rooms to minimize high‑pressure steam runs and heat losses. [everhealgroup]
When working with energy‑constrained or expansion‑ready sites, Everheal can:
- Place WFI generation skids near high‑consumption process blocks to shorten distribution loops and reduce heat loss. [everhealgroup]
- Reserve utility corridors and mezzanines for potential future membrane modules or hybrid upgrades. [everhealgroup]
- Coordinate HVAC and cleanroom zoning so that heat rejected from distillation or distribution loops can be partially recovered in other building systems. [everhealgroup]
These layout decisions directly affect CAPEX, lifecycle OPEX, and even the feasibility of future capacity expansion without major shutdowns. [austar.com]
Practical Decision Framework for Energy‑Constrained Facilities
In real projects, the decision between multi‑effect distillation and membrane‑based ambient WFI rarely comes down to a single factor. A practical, expert‑level framework typically includes the following steps: [wateronline]
1. Map utility constraints
Quantify available electrical, steam, and cooling capacity, as well as local fuel and electricity prices. [austar.com]
2. Define regulatory and quality expectations
Check local pharmacopeias and authority preferences, and evaluate your organization's comfort level with complex membrane control strategies. [csrwire]
3. Model lifecycle cost
Compare 15–20‑year scenarios including fuel, membrane replacement, major overhauls, and expected capacity expansions. [crbgroup]
4. Evaluate operational maturity
Assess whether your maintenance and QA teams can reliably manage ambient WFI monitoring and sanitization regimes. [wateronline]
5. Consider hybrid or phased solutions
Combine a smaller MED unit with membrane polishing or design space for a future ambient WFI line once energy infrastructure is upgraded. [wateronline]
Everheal's engineering teams routinely guide clients through this decision process, combining process simulations with on‑site utility surveys to recommend a balanced solution instead of a one‑size‑fits‑all technology. [everhealgroup]
Expert Perspectives: When Each Technology Makes Sense
From an industry‑expert perspective, there are clear scenarios where each approach is favored. [csrwire]
- Choose Multi‑Effect Distillation when:
- You are building in a market where regulators are more conservative or have limited experience with membrane WFI. [cy-water]
- You need hot WFI at the point of generation and want a technology that is inherently self‑sanitizing. [cy-water]
- Your site already has robust steam and cooling infrastructure, possibly supported by CHP or high‑efficiency boilers. [processandcontrolmag.co]
- Choose Membrane‑Based Ambient WFI when:
- Energy or water supply is constrained, expensive, or carbon‑priced. [crbgroup]
- You are designing a new biotech or vaccine facility with strong automation and data‑driven QA practices. [wateronline]
- Sustainability metrics and green‑building certifications are strategic priorities. [processandcontrolmag.co]
Everheal can also help clients deliberately design transitional strategies, such as installing MED today but reserving space, utilities, and piping routes for a future ambient WFI upgrade as regulations and organizational capabilities evolve. [everhealgroup]
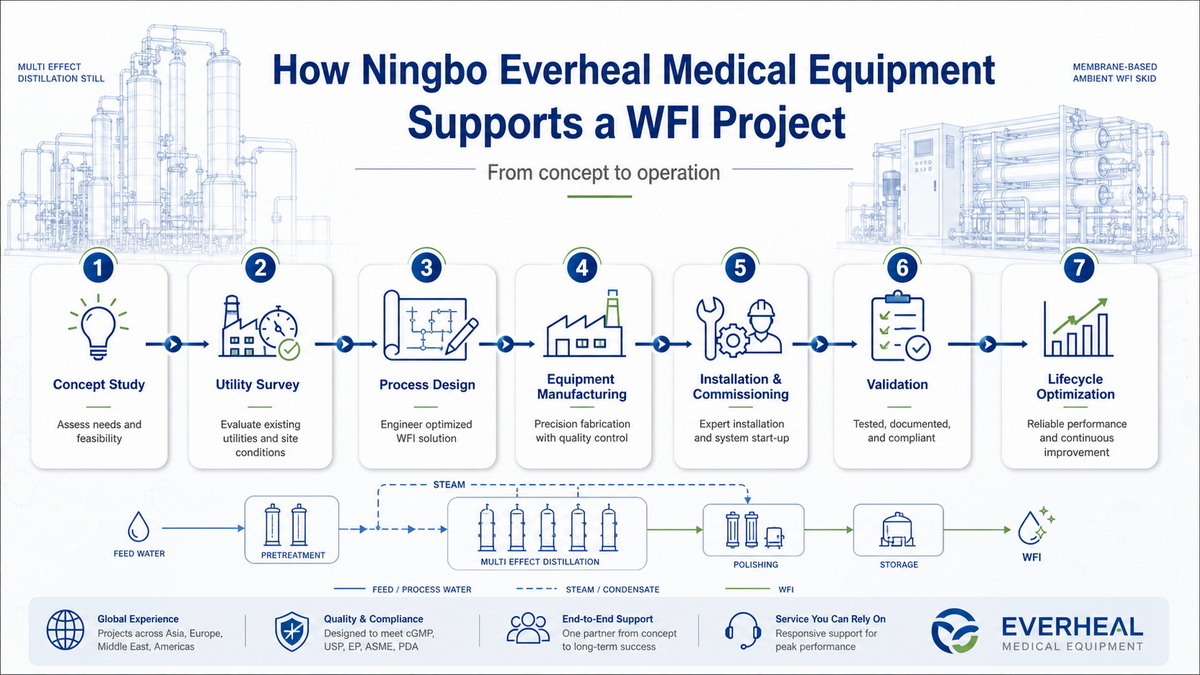
How Everheal Supports WFI Projects End‑to‑End
Ningbo Everheal Medical Equipment Co., LTD is a certified manufacturer of multi‑effect distillation water machines and related pharmaceutical utilities, with ISO‑based quality and environmental management systems. The company provides not only equipment but also complete factory layout planning, utility integration, and turnkey production‑line engineering for global pharma clients. [everhealgroup]
For energy‑constrained WFI projects, Everheal can:
- Design and manufacture multi‑effect distillation systems tailored to required WFI capacities and local utility conditions. [everhealgroup]
- Coordinate pure water, pure steam, WFI, and sterilization systems within a single, GMP‑aligned layout. [everhealgroup]
- Support commissioning, validation, and training so that operators can manage energy‑optimized WFI systems confidently over the full lifecycle. [everhealgroup]
A practical next step for many facilities is to engage Everheal with basic data on capacity, utilities, and regulatory targets, then iterate through a concept study comparing MED, membrane, and hybrid options. [everhealgroup]
Call to Action
If you are planning a new energy‑constrained facility or upgrading an older plant, align WFI generation with your long‑term energy, sustainability, and regulatory roadmap rather than treating it as a stand‑alone equipment decision. Contact Ningbo Everheal Medical Equipment Co., LTD to discuss a tailored WFI solution—multi‑effect distillation, membrane‑based ambient systems, or a hybrid architecture—integrated into a complete, GMP‑compliant factory layout. [everhealgroup]
FAQ
Q1. Is membrane‑based ambient WFI accepted by regulators worldwide?
Many major pharmacopeias now allow membrane‑based routes to WFI, but expectations around risk assessment, control strategies, and validation are strict and may vary by region. [csrwire]
Q2. Does multi‑effect distillation always consume more energy than membrane‑based WFI?
Multi‑effect distillation typically has higher steam and cooling demand, while membrane systems reduce these utilities but may use more electricity; the net impact depends on local energy prices and integration. [csrwire]
Q3. Can I retrofit an existing distillation‑based WFI system with membranes later?
Yes, many facilities design for a staged approach: install MED now, then add membrane‑based capacity or polishing skids as infrastructure, regulations, and budgets evolve. [wateronline]
Q4. What are the main operational risks of ambient WFI?
Key risks include microbial growth and biofilm formation at ambient temperatures, which require rigorous sanitization regimes, online monitoring, and strong QA oversight. [crbgroup]
Q5. How does Everheal support energy‑saving WFI projects?
Everheal combines equipment design with plant‑wide layout, utilities optimization, and GMP engineering services to deliver WFI solutions tailored to each facility's energy constraints and regulatory needs. [everhealgroup]
References
1. CRB, "Benefits of membrane filtration systems for ambient WFI production." [crbgroup]
2. WaterOnline / CSRwire, "Benefits of Membrane Filtration Systems for Ambient WFI Production." [csrwire]
3. Ningbo Everheal Medical Equipment Co., LTD – Corporate profile and high‑purity water generation solutions. [everhealgroup]
4. Everheal, "Multi Function Distillation Water Machine Supplier in China." [everhealgroup]
5. Everheal, "Pharmaceutical Filling Equipment – Integrated utilities and turnkey engineering." [everhealgroup]
6. Revolutionized / Process & Control, "Can modern innovations solve pharma's energy efficiency issues?" [processandcontrolmag.co]
7. AUSTAR, "Application of Building Energy‑Saving Technologies in the Pharmaceutical Industry." [austar.com]
8. CY‑Water, "注射用水系统WFI中多效蒸馏机技术解析." [cy-water]






















































