In GMP projects I support, the "pure steam vs. clean steam" question usually appears at URS or concept design stage, when the SIP philosophy is still flexible but early decisions will lock in validation strategy and lifecycle cost for 20+ years. As a China‑based system integrator, Ningbo Everheal Medical Equipment Co., LTD. increasingly sees global clients moving away from repurposed boiler steam toward dedicated pure steam generators for high‑risk aseptic and biotech lines, while using clean steam selectively for less critical utilities. [tian-sure]
What Do We Mean By Pure Steam And Clean Steam?
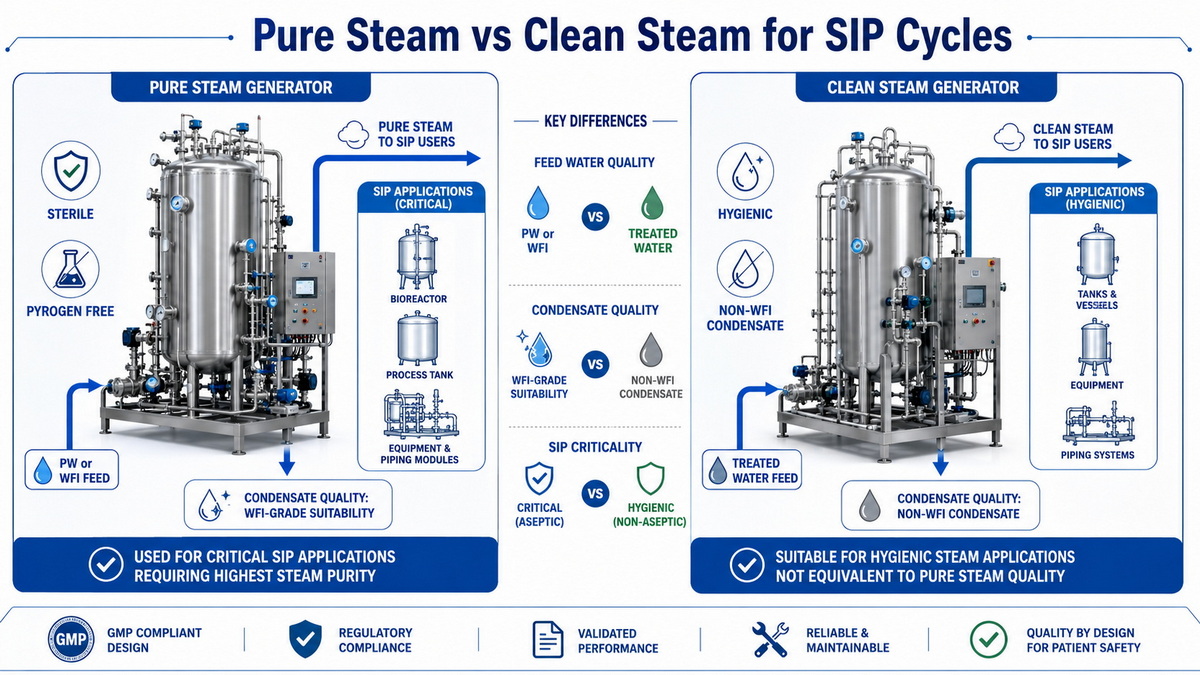
Pure Steam – Sterile, Pyrogen‑Free Steam For High‑Risk SIP
Pure steam is generated from purified water (often PW or WFI‑grade feed) in a dedicated pure steam generator designed to produce sterile, pyrogen‑free steam that condenses into condensate with quality close to WFI. Modern pharmaceutical pure steam generators use controlled evaporation and multi‑stage separation to remove droplets, particulates, non‑condensable gases, and potential contaminants before the steam reaches the sterilization load. [molewater]
Typical characteristics of pure steam for SIP: [pharmalab]
- Feedwater: purified water (PW) or WFI
- Steam quality: high dryness fraction, low non‑condensable gases
- Condensate quality: sterile, low endotoxin, low conductivity
- Use cases: SIP of product contact lines, sterile tanks, critical bioreactors, high‑risk aseptic processes
Clean Steam – Hygienic But Not Necessarily WFI‑Equivalent
Clean steam is usually generated from treated boiler feed water or purified water in a clean steam generator designed for hygienic applications, but its condensate is not always required to meet the same pharmacopeial limits as WFI. Clean steam systems are widely used in healthcare, food, and some pharmaceutical utilities, especially when the risk of direct product contact is lower. [wemacpharma]
Typical characteristics of clean steam: [tian-sure]
- Feedwater: demineralized or purified water, quality depends on application
- Steam quality: controlled for dryness and contaminants but not necessarily WFI‑grade condensate
- Use cases: sterilization of non‑product contact equipment, some HVAC humidification, certain CIP/SIP steps in lower‑risk areas
Key Technical Differences That Impact SIP Performance
From a SIP engineer's viewpoint, the real question is how these two steam types behave inside your piping, valves, and equipment during sterilization‑in‑place cycles. The table below summarizes the most important differentiators. [everhealgroup]
Technical Comparison For SIP Decision‑Making
| Aspect | Pure Steam Generators | Clean Steam Generators |
| Steam source | Dedicated pure steam generator using PW/WFI feed (everhealgroup) | Clean steam generator using treated feed water (tian-sure) |
| Condensate quality | Close to WFI, sterile and low endotoxin (everhealgroup) | Hygienic, but not always WFI‑equivalent (tian-sure) |
| Typical standards | Designed to support GMP sterile manufacturing, align with pharmacopeial expectations for pure steam (everhealgroup) | Often specified via internal clean utility standards; external pharmacopeias give broader guidance (tian-sure) |
| Primary use in SIP | High‑risk product contact SIP, sterile tanks, bioreactors, filling lines (everhealgroup) | Lower‑risk equipment, some non‑critical SIP loops, washer/disinfectors (tian-sure) |
| System complexity | More demanding in design, control, and validation (everhealgroup) | Simpler than full pure steam systems (tian-sure) |
| Typical cost | Higher capex and OPEX due to PW/WFI demand and equipment complexity (everhealgroup) | Moderate cost; can reuse some existing utilities (tian-sure) |
Because SIP validation focuses on reliable microbial kill, uniform temperature distribution, and repeatability, steam quality and condensate properties directly influence your biological indicator data and cycle robustness. In my experience, plants that compromise on steam quality often face recurring deviations in SIP mapping and longer troubleshooting cycles during qualification. [molewater]
Regulatory And GMP Expectations For SIP Steam
How Inspectors View Pure Steam Vs. Clean Steam
Regulators do not prescribe a single universal steam type for all SIP applications; instead, they expect a science‑ and risk‑based justification aligned with product criticality and contamination risk. For sterile injectables, vaccines, biotech APIs, and high‑risk parenterals, pure steam with WFI‑like condensate quality is widely accepted as the appropriate benchmark. [pharmalab]
Key inspection expectations typically include: [everhealgroup]
- Documented rationale for selecting pure steam or clean steam for each SIP user point
- Defined steam quality specifications (dryness, non‑condensable gas limits, superheat control)
- Routine monitoring of condensate quality at representative sampling points
- Clear linkage between steam utility design and product quality risk assessments
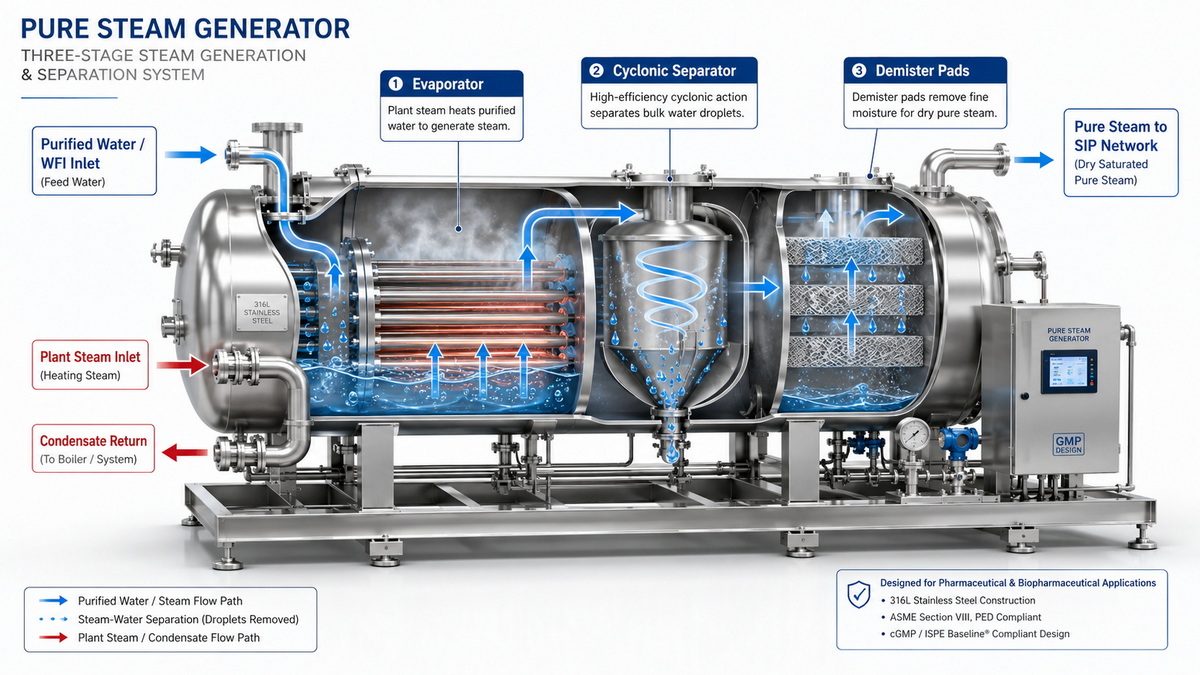
Impact On Validation And Documentation
Pure steam systems usually require more exhaustive documentation and qualification: detailed P&IDs, material certificates, welding records, FAT/SAT, and full IQ/OQ protocols. Clean steam systems may follow similar structure but often with less stringent condensate acceptance criteria and fewer sample points in lower‑risk zones. [wemacpharma]
For global buyers, Chinese manufacturers like Ningbo Everheal increasingly provide cGMP‑aligned documentation packages, remote FAT, and on‑site SAT/IQ/OQ support so that pure steam and clean steam systems can withstand overseas regulatory scrutiny. [everhealgroup]
SIP Cycle Reliability, Contamination Risk, And Lifecycle Cost
How Steam Quality Shapes SIP Outcomes
During SIP, poor steam quality can result in: [tian-sure]
- Cold spots due to wet steam and excessive condensate
- Longer come‑up times and cycle variability
- Chemical or particulate contamination from the steam source
- Difficulty achieving repeatable biological indicator kill rates
Pure steam generators, by design, target highly stable pressure and dryness, which reduces variability in temperature distribution and supports robust, shorter, and more predictable SIP cycles. Clean steam generators can also support reliable SIP when properly specified and maintained, but they leave less margin if your feedwater, piping, or load conditions fluctuate. [wemacpharma]
Total Cost Of Ownership (TCO) Over 15–20 Years
From a lifecycle cost point of view, pure steam often looks expensive at first but competitive over a 15–20‑year plant life when you factor in deviations, requalification, and contamination events. Plants that start with marginal steam quality sometimes face unplanned retrofits or separate pure steam generators later once products or regulations change, resulting in higher overall spend. [molewater]
Ningbo Everheal's turnkey projects show that combining pure steam generation with optimized plant layout, compact skids, and integrated clean utilities can rationalize pipe runs and reduce dead legs, which in turn lowers both CIP/SIP times and maintenance effort. [everhealgroup]
When To Specify A Pure Steam Generator For SIP
High‑Risk Scenarios Where Pure Steam Is The Default
Based on recent global projects and industry practice, pure steam generators are strongly preferred when: [pharmalab]
- Products are sterile injectables, vaccines, blood products, or high‑value biotech APIs
- SIP steam contacts product‑contact surfaces or sterile boundaries directly
- The facility targets alignment with stringent GMP regimes and international inspections
- Multiple SIP users (bioreactors, tanks, filling lines) demand tightly controlled and validated cycles
In these cases, using lower‑grade clean steam significantly increases the burden on justification and ongoing risk management.
Situations Where Clean Steam May Be Sufficient
Clean steam may be an acceptable, cost‑effective choice when: [tian-sure]
- Surfaces are not in direct contact with sterile product (e.g., some support equipment)
- Applications are in non‑critical zones or non‑parenteral dosage forms
- There is clear separation between clean steam and pure steam networks
- Your risk assessment demonstrates negligible impact on final product quality
Even then, I recommend harmonizing specifications so that clean steam quality still supports stable SIP thermodynamics and does not introduce avoidable particulate or chemical loads into equipment.
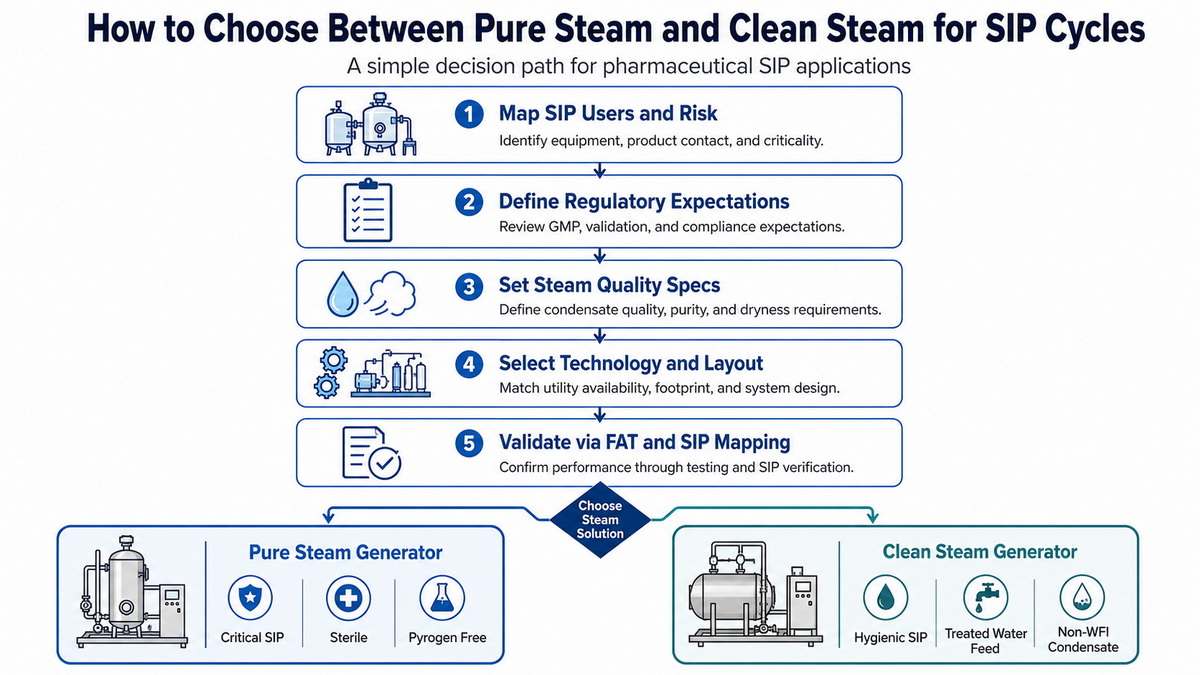
Expert Framework: Choosing The Right Standard For Your SIP Cycles
To make this decision less subjective, we often guide clients through a structured, 5‑step selection framework.
Step‑By‑Step Decision Process
1. Map SIP users and criticality
Classify each SIP user (tanks, process piping, filters, filling machines, isolators) by product contact and contamination risk. [everhealgroup]
2. Define regulatory and market expectations
Identify target markets and expected inspections (FDA, EMA, NMPA, WHO, etc.) and document applicable guidance on steam and water quality. [molewater]
3. Set steam quality specifications
For each risk class, define condensate limits, dryness fraction, non‑condensable gas limits, and acceptable superheat range. [pharmalab]
4. Select technology and layout
Decide where pure steam generators and clean steam generators should sit in the layout, how loops will be routed, and where reducers, condensate pots, and traps are placed to minimize dead legs. [everhealgroup]
5. Validate via modeling and FAT
Before shipment, conduct FAT with load simulations and SIP sequence checks; after installation, confirm by mapping, biological indicators, and condensate sampling. [everhealgroup]
This framework helps you justify when pure steam is non‑negotiable and where clean steam can be safely applied.
Case Insight: How A Mixed Pure Steam And Clean Steam Strategy Works In Practice
In recent years, international pharma and biotech companies have increasingly turned to Chinese pure steam generator manufacturers to support complex, mixed‑utility strategies. Ningbo Everheal, for example, acts not only as an equipment supplier but as a system integrator, combining pure steam generators, pure water systems, distillation units, and distribution loops into cohesive, validated packages. [everhealgroup]
A typical project approach includes: [everhealgroup]
- Jointly planning the water room and pure steam generator skids to minimize footprint
- Designing separate pure steam and clean steam lines, each with its own SIP users and sampling points
- Coordinating control philosophy with autoclaves, SIP systems, and cleanroom HVAC to avoid pressure shocks and cold spots
- Providing comprehensive documentation and support for FAT, SAT, IQ, and OQ
This allows clients to reserve pure steam for their most critical SIP cycles while still benefiting from clean steam in lower‑risk applications, all within a single, harmonized layout.
Practical Specification Tips For Project Teams
What To Include In Your URS
When drafting URS documents for pure steam generators or clean steam generators, make sure you clearly specify: [tian-sure]
- Required capacity and pressure range, based on simultaneous SIP loads and future expansion
- Feedwater quality (PW/WFI vs. demineralized water) and available utilities
- Steam quality and condensate acceptance criteria for SIP
- Materials of construction, surface finish, and welding standards
- Automation platform, alarms, and communication protocols with the plant DCS/SCADA
- Documentation requirements (P&IDs, wiring diagrams, certificates, FAT/SAT protocols)
Common Mistakes To Avoid
From both user and manufacturer side, we frequently see avoidable mistakes: [molewater]
- Underestimating peak SIP loads, leading to pressure drops and failed cycles
- Mixing pure steam and clean steam users without clear segregation or labelling
- Neglecting condensate sampling strategy and ongoing monitoring
- Treating steam quality as an "installation" topic instead of a lifecycle control parameter
Early collaboration with a pure steam generator manufacturer that understands GMP and international validation practice can prevent these issues.
How Ningbo Everheal Supports Pure And Clean Steam Decisions
Ningbo Everheal Medical Equipment Co., LTD. specializes in pure steam generators for pharmaceutical and biotech facilities and also supports integrated clean utility solutions. By combining engineering design, equipment manufacturing, and project execution, Everheal helps clients turn conceptual "pure vs. clean steam" discussions into practical, auditable solutions at layout and P&ID level. [everhealgroup]
Typical support includes: [everhealgroup]
- Early‑stage plant layout planning for pure water, WFI, and pure steam rooms
- Sizing and selection of pure steam generators tailored to SIP profiles
- Integration of pure steam generators with autoclaves, SIP loops, and filling lines
- End‑to‑end project support from URS review to FAT, SAT, IQ, and OQ
If your project requires more than a single utility skid, working with an integrator that understands both process and clean utilities will significantly reduce project risk.

Call To Action – Align Your SIP Steam Strategy With Your Next Project
If you are planning a new sterile facility, upgrading autoclaves, or re‑validating SIP loops, now is the right time to review whether pure steam or clean steam (or a combination) is truly aligned with your product risk and regulatory roadmap. Ningbo Everheal can support you with concept layout, URS review, and customized pure steam generator solutions that integrate smoothly into your existing or planned utilities, helping you build a robust, inspection‑ready SIP strategy from day one. [pharmalab]
Contact our engineering team to discuss your SIP loads, steam standards, and layout constraints, and we will help you design a pure steam or clean steam concept tailored to your plant.
FAQs: Pure Steam Generators, Clean Steam Generators, And SIP
Q1: Is pure steam always required for SIP?
Not always; pure steam is strongly recommended for high‑risk product contact SIP, but clean steam may be acceptable for lower‑risk equipment if justified by risk assessment and supported by data. [tian-sure]
Q2: Can I upgrade from clean steam to pure steam later?
Yes, but retrofits often require new generators, piping rearrangements, and re‑validation; it is usually more cost‑effective to design for pure steam at the start for critical lines. [molewater]
Q3: How do I size a pure steam generator for SIP?
Sizing should be based on the sum of maximum simultaneous steam demands from autoclaves, SIP circuits, sterilizable tanks, and other users, plus a safety margin for future expansion. [everhealgroup]
Q4: What tests prove steam quality for SIP?
Typical tests include dryness fraction, non‑condensable gas content, superheat checks, and condensate analysis for conductivity and endotoxins at representative sampling points. [pharmalab]
Q5: Can one generator serve both pure steam and clean steam users?
In principle, a properly designed pure steam generator can supply users that only require clean steam quality, but not vice versa; practically, many plants still implement separate networks for clarity and cost control. [tian-sure]
References
1. Everheal. "Top 10 Pure Steam Generator Manufacturers in China." [everhealgroup.com] [everhealgroup]
2. TianSure. "Pharmaceutical Pure Steam System | GMP Clean Steam Solution." [tian-sure.com] [tian-sure]
3. Everheal. "Pure Steam Generator Manufacturer – EVERHEAL." [everhealgroup.com] [everhealgroup]
4. Molewater. "纯蒸汽发生器对制药制造业至关重要." [molewater.com] [molewater]
5. Pharmalab. "Pure Steam Generation System." [pharmalab.com] [pharmalab]
6. WEMAC Pharma. "Applications of Clean Steam Generator for Sterilization in Healthcare." [wemacpharma.com] [wemacpharma]






















































