Pharmaceutical manufacturers building or upgrading high‑volume IV solution facilities face a strategic choice: invest in vapor compression (VC) distillation, or continue with multi‑effect distillation (MED) as the "default" WFI solution. As an equipment partner designing sterile process systems and complete production lines for global pharma clients, Everheal has seen how this single decision reshapes total cost of ownership, plant layout, and scalability over the full lifecycle of the asset. [linkedin]
From an engineering and operations perspective, lifecycle cost is no longer just about the purchase price of the still—it's the sum of energy, utility integration, maintenance, downtime risk, and future capacity expansions in an IV market that is expanding rapidly worldwide. [futuremarketinsights]
Why Lifecycle Cost Matters More in High-Volume IV Solution Production
The IV solutions and IV disposables markets are projected to grow at high compound annual growth rates (CAGR) over the next decade, driven by aging populations, higher procedure volumes, and chronic disease prevalence. For large‑scale IV solution plants, that means sustained, high, continuous demand for WFI and pure steam—often running 24/7 with limited downtime windows. [everhealgroup]
In this context, lifecycle cost includes:
- Capital expenditure (CAPEX) for VC or MED equipment and associated utilities. [aqua-chem]
- Operating expenditure (OPEX) for electricity, steam, cooling water, and pretreatment. [s20791.pcdn]
- Maintenance and spare parts over 15–20 years. [molewater]
- Downtime costs from unscheduled outages or difficult startups. [everhealgroup]
- Expansion costs when IV solution demand rises and WFI capacity must be scaled. [s20791.pcdn]
In high‑volume IV facilities, even small differences in kWh per cubic meter or steam usage translate into hundreds of thousands of dollars over the lifecycle of the plant. [peer.asee]
Technical Overview – Vapor Compression vs. Multi-Effect Distillation
How Vapor Compression Distillation Works
Vapor compression distillation uses a mechanical compressor (or thermocompressor) to recycle latent heat from the generated steam and feed it back into the evaporation process. Instead of continuously consuming high‑pressure plant steam, the VC unit compresses vapor, raising its temperature and pressure so it can serve as the heating medium for subsequent evaporation. [aqua-chem]
Key characteristics of VC systems for WFI:
- Higher energy efficiency because latent heat is reused rather than discarded. [molewater]
- Ability to generate ambient‑temperature WFI with lower additional cooling demand. [aqua-chem]
- Typically smaller footprint than comparable MED units for the same capacity. [meco]
- Simplified integration where electricity is reliable and competitively priced. [molewater]
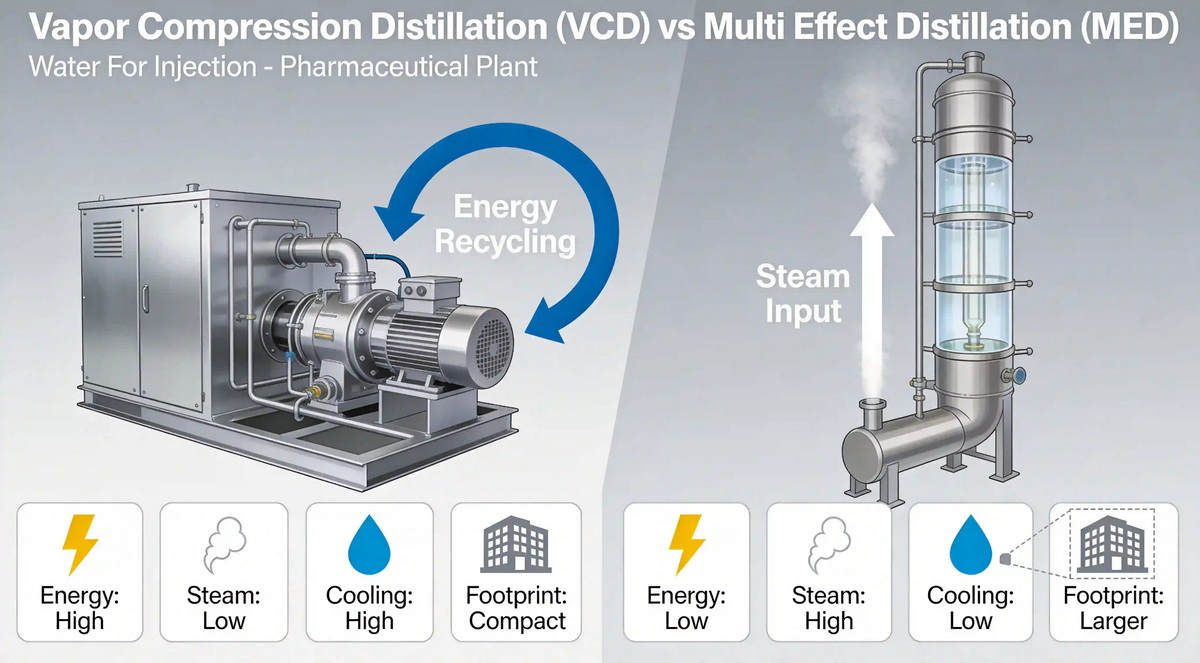
How Multi-Effect Distillers Operate
Multi‑effect distillers arrange several evaporation "effects" in series so that vapor from one stage serves as the heating medium for the next, improving thermal efficiency compared with single‑effect distillation. MED has been the traditional standard for WFI in many regulated markets and remains widely installed in both legacy and new facilities. [mhuanbao.bjx.com]
Key characteristics of MED systems:
- Driven primarily by plant steam as the main energy source. [s20791.pcdn]
- Strong track record and broad regulatory acceptance across major markets. [s20791.pcdn]
- High reliability in large installations, especially when combined with robust pretreatment. [sciencedirect]
- For ambient‑temperature WFI, MED often requires additional cooling, which increases total energy demand. [aqua-chem]
Comparative Lifecycle Cost Drivers in IV WFI Production
Energy Consumption and Utility Cost
Energy is the single largest driver of lifecycle cost for WFI systems in high‑volume IV production.
Evidence from desalination and WFI cost studies shows that VC and MED have broadly comparable energy ranges, but configuration and operating mode significantly affect the final kWh per cubic meter of product. In practice: [peer.asee]
- VC systems can achieve lower or comparable total cost because they leverage mechanical work (electricity) to recycle heat, especially where electricity prices are competitive. [molewater]
- MED units are tightly coupled to steam pricing, and their cost profile is more exposed to fluctuations in fuel prices and boiler efficiency. [aqua-chem]
A representative cost review for WFI production indicates: [s20791.pcdn]
- VC can produce ambient WFI at the lowest overall cost, because the system naturally condenses vapor into cooler WFI as part of the process. [aqua-chem]
- For MED, producing ambient WFI may actually cost more than producing hot WFI, due to extra cooling load. [aqua-chem]
For IV plants running large bag or bottle filling lines, where downstream processes may require controlled WFI temperatures, these differences in cooling and heating cycles significantly affect lifecycle cost.
Cooling, Pretreatment, and Ancillary Systems
Cooling and pretreatment are frequently underestimated in early project budgeting.
- In VC systems, feedwater cooling is used to re‑condense steam and recover energy, reducing the need for additional cooling utilities. [aqua-chem]
- MED often demands supplemental cooling capacity—chilled water or cooling tower load—especially when lower‑temperature WFI is required. [aqua-chem]
- For certain feedwaters with higher chloride content, reverse osmosis (RO) pretreatment may be required for MED, whereas VC can sometimes operate with less stringent pretreatment, depending on design and local water quality. [sciencedirect]
These ancillary utilities add to both CAPEX (cooling equipment, RO skids, water storage) and OPEX (chemicals, membrane replacement, pump power), and they scale with production volume and the number of operating shifts. [peer.asee]
CAPEX vs. OPEX Trade-Offs
From an investment perspective, pharma project teams often focus on the initial purchase price of the still. Yet cost reviews of WFI production show that the operational phase dominates total cost of ownership, especially beyond year five. [molewater]
General tendencies:
- MED may offer competitive or lower initial CAPEX, particularly in regions where suppliers and service networks are mature. [s20791.pcdn]
- VC may require higher initial investment for the compressor and controls but often delivers lower OPEX, especially in plants targeting ambient WFI or where steam is expensive. [molewater]
For a high‑volume IV solution facility expected to run at high utilization for 15–20 years, a slightly higher CAPEX for VC can be offset by substantial savings in energy and cooling costs over the lifecycle. [s20791.pcdn]
Operational Reliability, Maintenance, and Downtime Risk
Reliability Profiles in Continuous IV Production
Reliability data from thermal desalination and WFI applications shows that both MED and mechanical vapor compression technologies can achieve high system reliability, provided that pretreatment and operation are well managed. [sciencedirect]
For IV production lines, uptime is critical because:
- Any WFI system stoppage can halt formulation, compounding, and filling lines, leading to product loss and scheduling disruption. [everhealgroup]
- Restarts must consider sterilization-in-place (SIP), quality checks, and temperature stabilization.
MED systems benefit from a long operational history and well‑understood maintenance regimes in many pharmaceutical plants worldwide. VC systems, when properly engineered, offer high reliability as well, but place more emphasis on compressor health and vibration monitoring. [molewater]
Maintenance Complexity and Cost
Over the lifecycle of a WFI system, maintenance costs can become a significant portion of total OPEX.
- VC units have fewer evaporation stages but include a mechanical compressor that requires periodic inspection, lubrication, and possible overhaul. [molewater]
- MED units have multiple effects, each with heat transfer surfaces susceptible to scaling and fouling, requiring regular cleaning and inspection. [mhuanbao.bjx.com]
In practice, the cost balance depends on:
- Local availability of compressor service expertise versus experience with MED internals. [s20791.pcdn]
- Feedwater quality and fouling tendency.
- How well the plant's maintenance strategy is aligned with predictive monitoring and planned shutdowns.
For high‑volume IV plants with robust utility and maintenance teams, either technology can be managed effectively, but the maintenance cost profile should be included in lifecycle analysis alongside energy and cooling costs. [s20791.pcdn]
Footprint, Layout, and Scalability in IV Solution Plants
In greenfield projects, equipment footprint directly affects building size, cleanroom layout, and utility corridors.
VC systems typically offer a smaller footprint than equivalent MED units, which can free up space for additional IV filling lines, quality labs, or future capacity expansion. MED systems, with multiple effects, can require more height and floor area, influencing building design and steel structure costs. [meco]
From an Everheal plant‑layout perspective:
- VC‑based WFI systems can be integrated closer to high‑volume IV filling and compounding areas, with shorter WFI distribution loops and reduced heat loss. [linkedin]
- MED‑based WFI systems may be positioned with stronger links to central steam plants and cooling utilities, which can influence piping complexity and project timelines. [everhealgroup]
Scalability is another key factor. In fast‑growing IV markets, plant owners often plan phased expansions. VC systems can sometimes be scaled by adding parallel modules, while MED expansions may require more significant structural adjustments. [molewater]

Comparative Snapshot – VC vs. MED Lifecycle Cost Levers
Key Cost and Design Factors
| Factor |
Vapor Compression Distillation |
Multi-Effect Distillers |
| Primary energy source |
Electricity for compressor; lower steam demand. (molewater) |
Plant steam is primary driver of energy use. (aqua-chem) |
| Ambient WFI capability |
Produces ambient WFI with lower extra cooling cost. (aqua-chem) |
Ambient WFI often requires additional cooling, increasing OPEX. (aqua-chem) |
| Typical footprint |
Generally smaller footprint at similar capacity. (meco) |
Larger footprint and height due to multiple effects. (meco) |
| CAPEX trend |
Often higher initial CAPEX, especially for compressor and controls. (molewater) |
Frequently competitive CAPEX; long‑established baseline solution. (s20791.pcdn) |
| OPEX profile |
Favorable where electricity is competitive; lower total cost for ambient WFI. (molewater) |
OPEX strongly tied to steam and cooling costs. (aqua-chem) |
| Maintenance focus |
Compressor reliability, vibration, seals, bearings. (molewater) |
Heat transfer surface cleaning, scaling, multi‑effect balancing. (mhuanbao.bjx.com) |
| Regulatory track record |
Widely accepted where VC WFI systems are established. (molewater) |
Long global history as default WFI method. (s20791.pcdn) |
| Best fit use case |
High‑volume, long‑run IV plants targeting ambient WFI and space efficiency. (molewater) |
Plants with strong steam infrastructure and existing MED expertise. (everhealgroup) |
Expert Perspective – Choosing the Right Technology for High-Volume IV Plants
From the standpoint of a plant layout and equipment integrator, the right choice is contextual, not dogmatic. Based on Everheal's experience designing complete sterile production lines, the decision typically hinges on four questions: [linkedin]
1. What is your target WFI temperature at points of use—hot, ambient, or both?
2. How are electricity and steam priced and generated in your region?
3. How aggressively will your IV production volume grow over the next 10–15 years?
4. What is the maturity of your maintenance and utility teams for thermal systems and compressors?
In many high‑volume IV solution projects today, VC emerges as the preferred option when:
- IV solutions are filled at large scale with high demand for ambient‑temperature WFI. [aqua-chem]
- The site has reliable electrical infrastructure and wants to reduce dependence on steam. [molewater]
- Space is at a premium, and a compact WFI system unlocks more floor area for production lines. [meco]
MED remains a strong, defendable choice where:
- Existing plants already rely on MED infrastructure and operators are highly familiar with the technology. [s20791.pcdn]
- Steam is inexpensive and abundantly available from efficient central boiler plants. [aqua-chem]
- Regulatory or corporate preferences favor established, long‑running MED designs.
Practical Lifecycle Cost Evaluation Framework (Step-by-Step)
A Structured Approach for Project Teams
To move from high‑level comparisons to a project‑specific decision, Everheal typically guides IV manufacturers through a structured evaluation:
1. Define capacity and profiles
- Daily and peak WFI demand, hot vs. ambient temperature split, and IV product mix (bags, bottles, pre‑mixed solutions). [everhealgroup]
2. Map utility prices and availability
- Current and projected electricity and steam costs, boiler efficiency, and cooling water constraints. [s20791.pcdn]
3. Model CAPEX and OPEX over 15–20 years
- Include equipment, utilities, pretreatment, maintenance, and expansion scenarios for both VC and MED. [molewater]
4. Simulate plant layout and space usage
- Compare footprints, access space, cleanroom boundaries, and future expansion zones under VC and MED options. [meco]
5. Assess operational risk and redundancy
- Evaluate single vs. multiple trains, standby capacity, and maintenance strategy to ensure IV production continuity. [peer.asee]
Using this framework, plant owners gain a quantitative view of lifecycle cost, not just vendor quotes, and can align the WFI strategy with long‑term IV market demand.
Example Scenario – High-Volume IV Solution Facility
Consider a greenfield IV solution plant with:
- High‑speed form‑fill‑seal lines for large‑volume parenterals.
- 24/7 operation with minimal planned downtime.
- Requirement for both hot and ambient WFI at multiple points of use.
In this scenario:
- A VC‑based system can provide ambient WFI at lower overall cost, with reduced cooling load and strong lifecycle economics when electricity pricing is competitive. [aqua-chem]
- MED may still be attractive if the site already has an oversized, efficient steam plant and if corporate engineering standards favor MED, but its additional cooling requirements for ambient WFI will raise OPEX over time. [s20791.pcdn]
From an integrator's point of view, many IV projects increasingly favor VC for new high‑volume lines, while retaining MED in legacy plants or hybrid configurations where both technologies coexist based on local constraints. [everhealgroup]
How Everheal Supports Your VC vs. MED Decision
As an integrated partner, Everheal provides end‑to‑end engineering support for sterile manufacturing and IV solution plants, from process design to commissioning. This includes: [linkedin]
- Feasibility studies comparing VC and MED for specific markets and utility scenarios. [molewater]
- Customized plant layout design ensuring optimal footprint, flows, and utility integration. [linkedin]
- Delivery of multi‑effect water distillation machines, pure steam generators, and complementary sterile process equipment as a coordinated package. [everhealgroup]
By combining equipment manufacturing with process engineering and layout planning, Everheal helps IV manufacturers implement a lifecycle‑optimized WFI strategy that aligns with long‑term demand and regulatory expectations.

Call to Action – Plan Your WFI Lifecycle Cost Study
If you are planning a new high‑volume IV solution plant or upgrading legacy WFI infrastructure, this is the right time to quantify the lifecycle cost difference between vapor compression distillation and multi‑effect distillers in your specific context.
Everheal's engineering team can support you with:
- A data‑driven VC vs. MED lifecycle cost model tailored to your utility prices and IV demand forecasts.
- Conceptual plant layout options showcasing footprint, scalability, and integration trade‑offs.
- A complete, compliant equipment package designed around your preferred WFI technology.
Contact Everheal to discuss your project requirements and schedule a VC vs. MED lifecycle cost assessment for your next IV solution investment.
FAQs
1. Is vapor compression distillation always cheaper than multi-effect distillation?
Not always. VC tends to offer lower or comparable total cost where electricity is competitively priced and ambient WFI is required, but MED can remain cost‑effective in plants with abundant low‑cost steam and existing MED expertise. [aqua-chem]
2. Are both VC and MED acceptable for WFI under major regulations?
Yes. Both VC and MED technologies are widely used for producing WFI in regulated markets, provided they are properly designed, validated, and operated according to current guidelines and pharmacopeial requirements. [s20791.pcdn]
3. How does the choice of VC vs. MED affect IV solution quality?
When systems are correctly designed and validated, both VC and MED can consistently meet WFI quality specifications, so the impact on IV solution quality is driven more by overall system integration, storage, and distribution strategy than by the distillation method itself. [everhealgroup]
4. Can I switch from MED to VC in an existing IV plant?
Yes, but it requires careful engineering. Retrofitting VC into an existing plant involves reassessing utilities, distribution loops, available footprint, and validation impact. Many manufacturers choose a phased approach or hybrid setups during major upgrades. [everhealgroup]
5. What is the typical payback period for switching to VC?
The payback period depends on local energy costs, production volume, and whether ambient WFI is required. In high‑volume IV plants with high steam costs, the savings from VC can offset higher CAPEX over the medium term, often within the lifecycle planning horizon of the facility. [molewater]
References
1. MECO. "Vapor Compression vs. Multiple Effect Distillation." [Link]. [meco]
2. Future Market Insights. "IV Disposables Market – Global Market Analysis Report."[Link]. [futuremarketinsights]
3. Molewater. "Hybrid WFI System vs. Conventional Distillation: Cost Comparison." [Link]. [molewater]
4. Aqua‑Chem. "Vapor Compression Distillation vs Multiple Effect Distillation – White Paper." [PDF]. [aqua-chem]
5. MECO. "WFI: A Cost Review – A Cost Evaluation of Alternative Systems." [PDF]. [s20791.pcdn]
6. Environmental Impact and Cost Analysis of Multi‑Stage Flash, Multi‑Effect Distillation, Mechanical Vapor Compression, and Reverse Osmosis. ASEE paper. [peer.asee]
7. Everheal Group. Company and product information – pharmaceutical sterile process systems and water systems.[Link]. [linkedin]
8. Fortune Business Insights. "Intravenous (IV) Equipment Market Size, Share and Industry Analysis."[Link]. [fortunebusinessinsights]
9. Investigation of multi‑effect mechanical vapor compression systems – ScienceDirect abstract and related literature on energy performance and reliability. [sciencedirect]






















































