Glass bottles and flexible IV bags each have clear strengths in filling efficiency and sterilization stability, and the best choice depends on your product profile, regulatory strategy, and plant layout roadmap. For manufacturers investing in new sterile IV lines, combining modern BFS/FFS technologies with packaging choice is often the real competitive lever, rather than treating "glass vs. bags" as a simple either–or. [rommelag]
Glass Bottles vs. Flexible IV Bags: Why the Debate Matters
In large‑volume parenteral (LVP) and IV solution production, the packaging format shapes everything from line speed and yield to contamination risk and lifecycle cost. Glass bottles remain the gold standard for chemical stability and certain high‑risk formulations, while flexible IV bags dominate in logistics efficiency and patient handling. [aipakengineering]
From an equipment perspective, traditional glass bottle lines rely on depyrogenation tunnels and multi‑machine layouts, whereas flexible IV bags are increasingly produced on integrated FFS (Form‑Fill‑Seal) and BFS (Blow‑Fill‑Seal) systems that compress forming, filling, and sealing into one aseptic platform. For a manufacturer like Ningbo Everheal Medical Equipment that designs both FFS and BFS lines, this comparison is not theoretical—it determines conveyor layouts, cleanroom zoning, and long‑term OEE (Overall Equipment Effectiveness). [soontact]
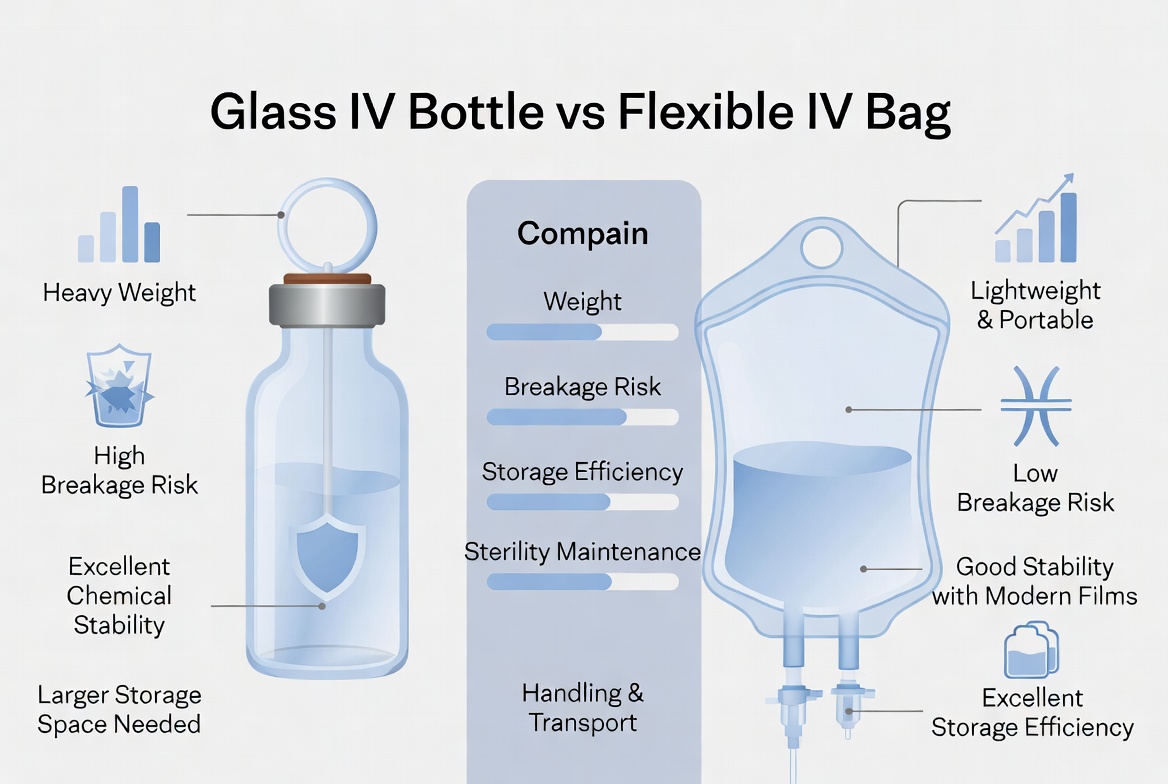
Core Technical Differences Between Glass Bottles and Flexible IV Bags
Material Properties and Chemical Interaction
Glass offers excellent chemical inertness and gas barrier performance, which is why it remains preferred for sensitive or highly reactive drug products and certain high‑end formulations. Borosilicate glass in particular has a long clinical track record and clear regulatory acceptance in parenteral use. However, glass is brittle, heavy, and vulnerable to delamination and breakage, which introduces particulate risk and adds cost to handling and secondary packaging systems. [canadianmedhealthsupplies]
Flexible IV bags, typically made from polyolefin or multilayer films (such as PP‑ or PE‑based structures), provide good compatibility for many aqueous solutions and enable lightweight, collapsible containers that simplify infusion and reduce storage volume. Modern films are engineered to balance gas barrier, leachables control, and weldability so they can be aseptically formed and sealed on FFS and BFS lines. That said, the presence of polymers introduces concerns about extractables, leachables, and the newer issue of microplastic release into IV fluids, which regulators and hospitals are watching more closely. [acs]
Container Geometry and Headspace
Glass bottles are rigid, with defined headspace and internal geometry, supporting precise control of fill volume and over‑fill margins on high‑speed rotary fillers. The rigid shape aids in visual inspection but demands vented sets or special administration devices to avoid vacuum issues during infusion. [zoledronicpremix]
Flexible IV bags, in contrast, are collapsible, which allows gravity infusion without air venting and reduces the risk of air embolism when used correctly. Their rectangular or flat design improves palletization efficiency and shelf density in hospital pharmacies. For equipment engineers, bag geometry must be considered in the forming station design, web tension control, and seal tooling of FFS systems to ensure consistent chamber evacuation, sealing, and hanging‑hole integrity. [zoledronicpremix]
Filling Line Efficiency – Throughput, Layout, and OEE
Throughput and Cycle Times
Modern plastic IV production lines reach very high nominal capacities: advanced BFS and FFS systems for plastic containers can achieve outputs in the tens of thousands of bottles or bags per hour, depending on configuration and container volume. By comparison, high‑end glass bottle IV lines may reach around 450 bottles per minute (roughly 27,000 bottles per hour), but this often requires a series of specialized machines and larger footprint. [pharmaguideline]
In practice, effective throughput is shaped by changeover times, sterilization cycles, and reject rates. BFS technology forms the container, fills, and seals in a single aseptic chamber in 10–15 seconds per mold set, with minimal human intervention, which significantly lowers micro‑stoppages and contamination events. FFS bag lines, especially vertical FFS systems, can also run at 100–200 packages per minute per lane, and multi‑lane execution further multiplies output. [soontact]
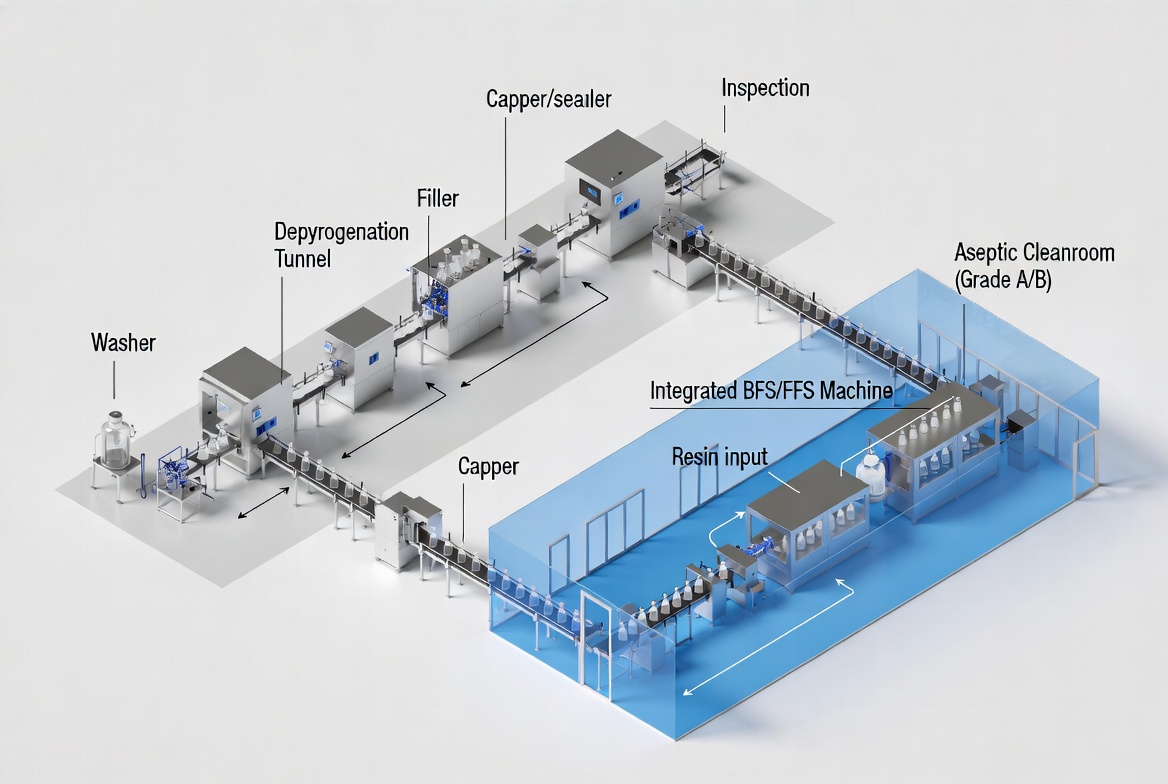
Line Architecture and Footprint
Glass bottle lines typically combine separate equipment modules—washer, depyrogenation tunnel, filler, capper, inspection, and sometimes external sterilization—connected by conveyors and complex transfer points. This architecture increases line length and requires larger cleanroom areas and HVAC capacity. [pharmaguideline]
BFS systems are compact, self‑contained units in which extrusion, blowing, filling, and sealing happen inside one enclosed machine, often occupying 100–200 square feet for the core process. FFS lines for flexible bags have a longer web travel path but can still be arranged more compactly than traditional bottle lines with equivalent output, especially when Everheal's engineering team optimizes upstream film handling and downstream inspection and cartoning in a unified layout. [rommelag]
Automation, Labor, and Changeover
Glass bottle filling typically involves more manual interventions: bottle loading, format parts changes, and more frequent adjustments to accommodate different heights, neck finishes, and closures. Each human touchpoint in an ISO‑classified area adds both labor cost and contamination risk. [aipakengineering]
In contrast, BFS and FFS lines rely on recipe‑driven parameters and quick‑change tooling, and they can integrate CIP/SIP (clean‑in‑place/sterilize‑in‑place) routines for product contact parts. For pharmaceutical manufacturers running multiple SKUs, flexible bag lines can be engineered for faster format changeovers by standardizing port designs and bag formats, a design strategy where an experienced OEM can add real value during conceptual layout. [soontact]
Sterilization Stability and Aseptic Assurance
Inherent Sterility and Process Design
Traditional glass bottle processes rely on external sterilization: containers are cleaned and depyrogenated in a tunnel at high temperature, then transported to a classified filling environment. Each transfer step between washer, tunnel, and filler is controlled but still represents a point where airborne contaminants or mechanical defects can occur. [pharmaguideline]
BFS is considered an inherently aseptic technology by regulators because the container's interior is created from hot plastic within a closed chamber, immediately filled and sealed under sterile conditions. The combination of high melt temperature and enclosed environment minimizes bioburden and virtually eliminates operator intervention during the critical process steps. Aseptic FFS systems for IV bags also provide strong sterility assurance when combined with film sterilization (e.g., gamma or hydrogen peroxide treatment) and validated aseptic barriers, though they are not as intrinsically aseptic as BFS. [rommelag]
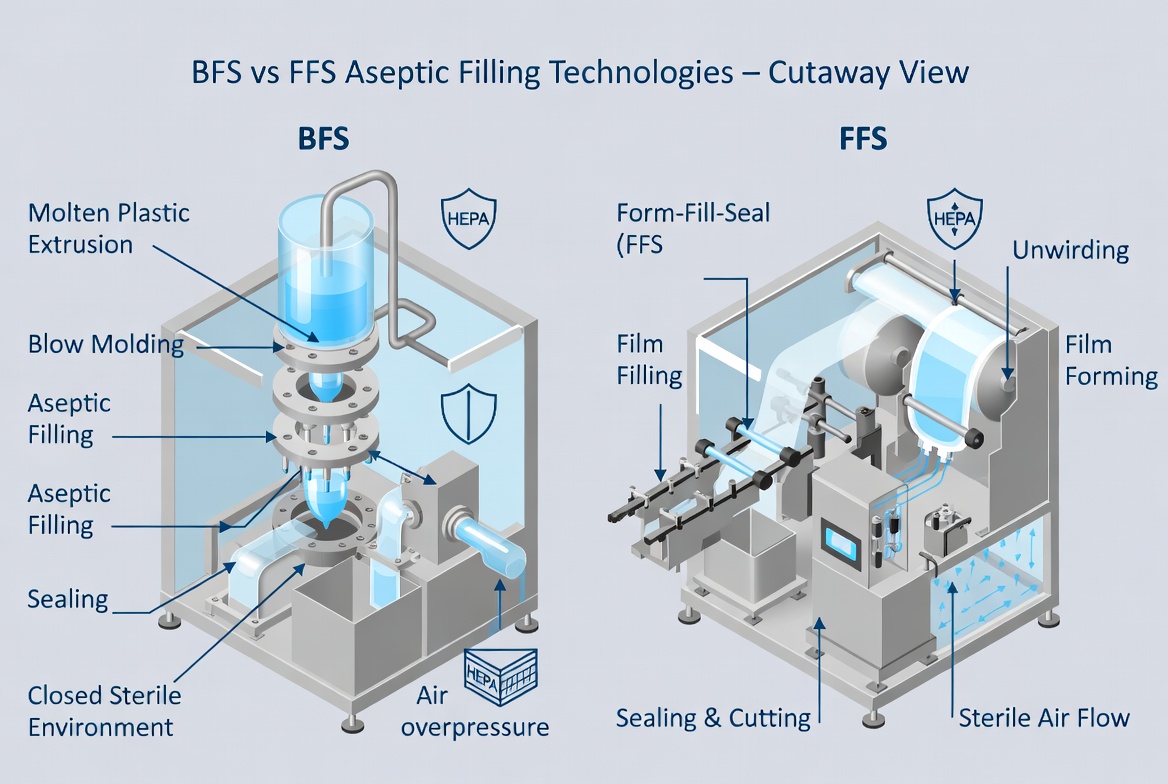
Long‑Term Stability and Container–Closure Integrity
Glass provides excellent long‑term chemical stability and low gas permeability, supporting multi‑year shelf lives for many solutions when combined with appropriate closures. However, glass is susceptible to micro‑cracks, delamination, and particulate release, especially when subjected to thermal shocks or mechanical stress in high‑speed lines. These issues can compromise container–closure integrity and lead to costly recalls. [canadianmedhealthsupplies]
Flexible IV bags rely on film integrity and seal quality for their barrier performance. Multilayer films have been developed specifically to support long shelf life of premixed IV drugs in the US and EU markets, with improved resistance to oxygen ingress and sorption. At the same time, recent studies report that plastic IV bags can shed microplastics into infusion solutions, delivering thousands of particles per 250‑ml bag, which raises new questions for toxicology and risk assessments. For a plant planning new lines, this means that film selection and supplier qualification must be treated as a critical path task alongside equipment choice. [pmc.ncbi.nlm.nih]
Microbiological and Particulate Risks
With glass bottles, particulate contamination risk often comes from glass flakes, closures, and line components, even when sterilization is robust. Strict visual inspection and automated particle detection are therefore mandatory. [aipakengineering]
Flexible bags avoid glass particulates and usually yield fewer breakage‑related rejects, but introduce polymer‑derived particles and microplastics, particularly under UV exposure or heat. Good practice now includes controlling storage conditions for empty bags and filled products—keeping them away from high temperatures and UV light to reduce microplastic shedding. BFS technology, by integrating container formation and filling in one closed step, can significantly reduce both microbial and particulate risks compared to traditional bottle filling. [acs]
Operational and Economic Considerations for IV Manufacturers
Cost per Unit and Total Cost of Ownership
Glass bottle packaging incurs costs for bottle purchase, transport, depyrogenation energy, and breakage‑related waste. While glass itself is relatively inexpensive, the overall logistics and handling burden is high. [aipakengineering]
Plastic IV bags on FFS or BFS lines eliminate the need for pre‑made containers; BFS uses raw resin, and FFS uses rollstock film, both of which can reduce packaging costs per unit and lower warehouse space needs. Equipment CAPEX can be higher for BFS, but OPEX benefits—simplified logistics, lower labor, and higher yield—often offset this over the system's lifecycle. An OEM with both BFS and FFS expertise can model payback scenarios based on SKU mix, regional wage levels, and utility costs to support investment decisions. [rommelag]
Sustainability and Environmental Impact
Glass is fully recyclable and widely accepted in existing recycling streams, which supports hospital sustainability initiatives, but its weight increases transport emissions. [canadianmedhealthsupplies]
Plastic IV bags are lighter and reduce shipping‑related emissions and storage footprints, and BFS in particular forms containers to exact size, minimizing excess material. However, recycling of medical plastics is challenging, and concerns about microplastics in the bloodstream highlight the need for next‑generation films and better post‑use waste management. For some hospitals and regulators, the future may lie in hybrid strategies—maintaining glass for the most sensitive drugs while shifting standard electrolytes and hydration solutions to optimized plastic bag systems with improved recyclability. [journals.sagepub]
Expert Perspective – Matching Packaging to Process and Product
From a process engineering and layout‑planning perspective, the "best" choice is rarely glass or bags in isolation; it is a packaging + line + facility combination. Experienced line designers will start from:
- Product characteristics (pH, osmolarity, sensitivity to oxygen/light)
- Required shelf life and distribution chain conditions
- Regulatory positioning (BFS as advanced aseptic technology)
- Target throughput and SKU complexity
Then they map these requirements to either:
- A high‑speed, inspection‑intensive glass bottle line with strong depyrogenation and visual control, or
- An FFS or BFS platform for flexible bags or plastic containers with simplified aseptic zoning and higher integration. [soontact]
In practice, many global manufacturers operate mixed portfolios: glass for certain injectables and niche formulations, and flexible IV bags for high‑volume saline, dextrose, and electrolyte solutions. For new greenfield factories, early collaboration with equipment suppliers allows building a layout that can support both technologies with shared utilities and modular expansion options. [canadianmedhealthsupplies]
Case‑Style Scenario – Upgrading from Glass to Flexible IV Bags
Consider a regional hospital‑supply manufacturer currently running a 300‑bpm glass bottle line for LVPs and facing frequent downtime due to bottle breakage and tunnel maintenance. By introducing a BFS or FFS line for flexible IV bags, they could: [pharmaguideline]
1. Reduce bottle‑handling issues and associated rejects.
2. Increase pallet density and cut logistics costs due to lighter, flat‑packable bags. [zoledronicpremix]
3. Improve aseptic assurance through closed‑system forming and filling, especially with BFS. [soontact]
4. Offer premixed bag formats that may support lower infection rates compared with multi‑step admixture processes. [zoledronicpremix]
However, they would also need to:
- Qualify new films and ports from approved suppliers. [journals.sagepub]
- Address microplastic risk via controlled storage and film selection. [pmc.ncbi.nlm.nih]
- Re‑validate shelf‑life and stability studies in the new container format. [journals.sagepub]
An experienced OEM and layout partner can guide these trade‑offs, leveraging prior project data to shorten validation timelines and de‑risk the transition.
Practical Guidance for Choosing Glass Bottles or Flexible IV Bags
For decision‑makers planning new or upgraded lines, the key considerations can be summarized as follows.
Decision Factors Table
| Factor | Glass Bottles | Flexible IV Bags (FFS/BFS) |
| Chemical stability | Very high for many formulations (canadianmedhealthsupplies) | Good with modern films; depends on film design (journals.sagepub) |
| Aseptic assurance | Strong but multi‑step; more transfers (pharmaguideline) | BFS inherently aseptic; FFS can be highly aseptic (soontact) |
| Throughput potential | High; up to ~450 bpm on advanced lines (aipakengineering) | Very high; tens of thousands units/hour possible (aipakengineering) |
| Breakage and particulates | Breakage risk; glass particles possible (aipakengineering) | No glass breakage; microplastics concern (acs) |
| Logistics & storage | Heavy, bulky, more space needed (zoledronicpremix) | Lightweight, compact, easier storage (zoledronicpremix) |
| CAPEX for line | Moderate to high, multiple machines (aipakengineering) | Higher for BFS, variable for FFS; more integrated (soontact) |
| OPEX & labor | Higher labor, more manual handling (pharmaguideline) | Lower labor, more automation (soontact) |
| Sustainability | Recyclable but heavy (canadianmedhealthsupplies) | Lightweight, material recycling more complex (acs) |
| Regulatory perception | Long history, well understood (canadianmedhealthsupplies) | BFS recognized as advanced aseptic by FDA (soontact) |
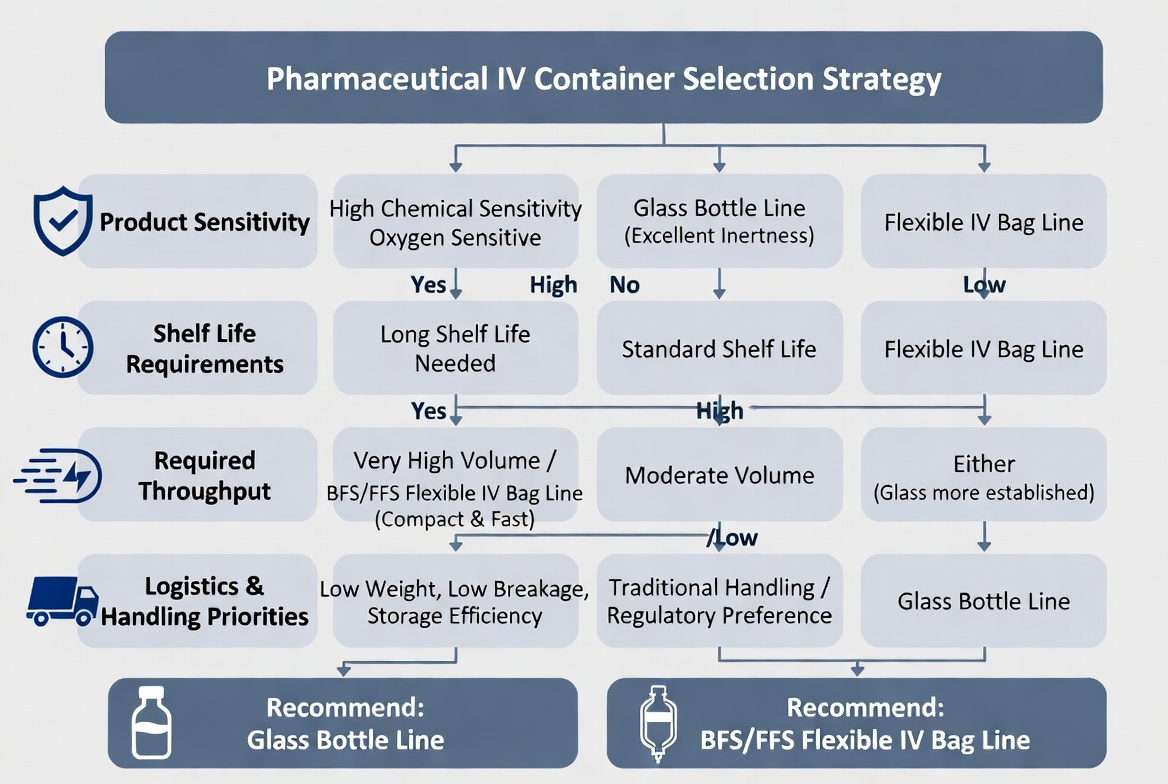
Where FFS and BFS Lines Add Strategic Value
For a manufacturer of GMP‑compliant IV solutions, the choice of FFS vs. BFS is as strategic as the choice of glass vs. bags. BFS excels when:
- Sterility risk must be minimized and the product is liquid only. [pharmaguideline]
- You want compact, highly integrated systems that reduce cleanroom footprint and human intervention. [soontact]
FFS is attractive when:
- You need flexible bag formats, multi‑chamber designs, or specific port/hanger geometries. [rommelag]
- You plan to run multiple SKUs and wish to leverage film‑based customization.
By combining BFS for critical aseptic products with FFS for customized flexible IV bags, a plant can cover a wide portfolio with harmonized maintenance, training, and spare‑parts strategies. An OEM that designs both machine types and supports layout planning can ensure utilities, cleanroom zoning, and downstream packaging equipment are aligned from day one.
Call to Action – Planning Your Next IV Filling Line
If you are evaluating glass bottle lines vs. flexible IV bag lines for a new project or retrofit, the most effective next step is a structured layout and process review. By sharing your current product list, target capacities, and regulatory markets, you can obtain a tailored concept that maps each SKU to the optimal combination of packaging type (glass vs. bag) and line technology (BFS vs. FFS).
A specialist equipment partner that delivers both FFS and BFS platforms, as well as end‑to‑end factory layout planning, can help you quantify throughput, sterility risk, and total cost of ownership before you commit to major CAPEX. Aligning on this early will turn the "glass bottles vs. flexible IV bags" debate into a clear, data‑backed roadmap for your next‑generation IV production facility.
FAQs
Q1: Are flexible IV bags always more efficient than glass bottles?
A: Flexible bags on FFS or BFS lines often deliver higher throughput and lower logistics costs, but actual efficiency depends on product mix, automation level, and layout design. [aipakengineering]
Q2: Do glass bottles offer better sterility than IV bags?
A: Glass bottles can achieve excellent sterility when combined with proper depyrogenation and aseptic filling, but BFS containers have an inherent aseptic advantage because they are formed, filled, and sealed in one closed step. [pharmaguideline]
Q3: How serious is the microplastic issue in IV bags?
A: Recent studies have detected thousands of microplastic particles in single IV bags, prompting calls for better film design and controlled storage away from heat and UV, though clinical impact is still being investigated. [acs]
Q4: When should I choose glass bottles for my IV product?
A: Glass is often chosen for highly sensitive formulations, drugs with long shelf life requirements, or when regulators or customers specifically prefer traditional glass packaging. [journals.sagepub]
Q5: Can one facility run both glass and flexible IV bag lines efficiently?
A: Yes, many manufacturers operate mixed portfolios, and a carefully planned layout with shared utilities and harmonized downstream packaging can support both technologies efficiently. [aipakengineering]
References
1. Aipak Engineering. "Glass Bottle IV Solution Production Line vs. Plastic." 2026. [aipakengineering]
2. Canadian Med Health Supplies. "Glass vs. Plastic: Which is Better for Medical Packaging?" 2025. [canadianmedhealthsupplies]
3. Rommelag. "Form Fill Seal: Aseptic filling of pharmaceuticals." 2026. [rommelag]
4. Soontact. "What Is The Difference Between Blow Fill Seal (BFS) And Form Fill Seal (FFS)?" 2025. [soontact]
5. Pharmaguideline. "Blow Fill Seal (BFS) and Form Fill Seal (FFS) Technology in Sterile Production." 2024. [pharmaguideline]
6. ACS / Environment & Health. "Medical infusion bags can release microplastics, study shows." 2025. [acs]
7. PMC. "Health Risks from Microplastics in Intravenous Infusions." 2025. [pmc.ncbi.nlm.nih]
8. Zoledronic Premix. "Premix Bags vs Ready-to-use Glass Bottles." 2025. [zoledronicpremix]
9. SAGE Journals. "Development of a multilayer film for the first US Food and Drug Administration–approved IV premixed drug product." 2024. [journals.sagepub]






















































