For biopharma teams, choosing between BFS and FFS is no longer just a packaging decision; it is a *risk management* decision that directly affects patient safety, batch yield, and regulatory confidence. From my experience working with CDMOs and pharma plants in Asia and Europe, the most successful projects are those that align the container–closure system with the *thermal sensitivity profile* of the biologic and the contamination risk tolerance of the target market. [vxpbiologics]
In this article, we will compare BFS and FFS through the lens of aseptic assurance, contamination pathways, and real-world implementation for thermolabile biologics, then share practical selection checklists and layout tips based on large-scale BFS/FFS lines similar to those delivered by Ningbo Everheal Medical Equipment Co., LTD. [rommelag]
Core Principles of BFS and FFS in Biologic Fill-Finish
BFS and FFS both integrate forming, filling, and sealing in one line, but they *control contamination* in very different ways. Understanding these fundamentals is essential before you assess which one better protects thermosensitive APIs, proteins, and vaccines. [susupport]
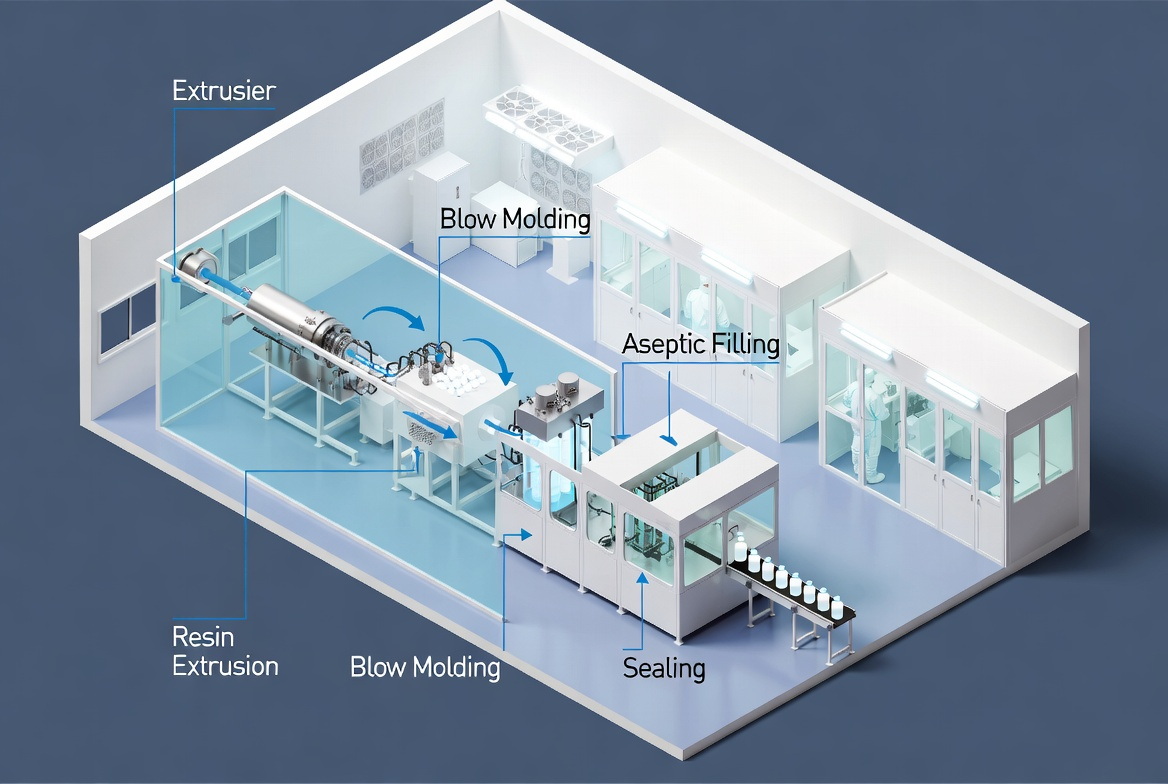
What Is Blow-Fill-Seal (BFS)?
In a BFS system, the machine extrudes molten thermoplastic resin (often LDPE, HDPE, or PP) into a hot parison, blow-molds the container, fills it with sterile product, and seals it in a fully enclosed, aseptic chamber in one continuous cycle. These cycles typically complete in under 20 seconds, which dramatically limits environmental exposure time for the biologic. Because the container is formed while the plastic is at high temperature inside a controlled environment, the container body itself gains a degree of inherent sterility before contact with product. [apiject]
Key aseptic advantages of BFS: [linkedin]
- Closed system from container formation through sealing, with minimal or no open transfer.
- Very limited human intervention during critical aseptic steps.
- High process repeatability thanks to full automation.
- Strong regulatory recognition as an advanced aseptic technology by authorities like the FDA. [soontact]
What Is Form-Fill-Seal (FFS)?
In FFS, the equipment forms flexible or semi-rigid packages from pharmaceutical-grade film, fills them with product, and heat-seals them along the line. The film is unwound, shaped into a pouch, sachet, or bag, then filled and sealed, often within a controlled environment using decontamination steps such as hydrogen peroxide (H₂O₂) or steam treatment of the film or forming area. [rommelag]
Key aseptic characteristics of FFS: [linkedin]
- More open film path, so the material can be exposed to ambient air for longer distances and times.
- Requires additional sterilization and decontamination steps to achieve high aseptic assurance (e.g., sterilant sprays, laminar airflow, isolators).
- Very flexible in package formats and suitable for liquids, powders, and some devices.
Contamination Risk Pathways: BFS vs. FFS for Thermosensitive Biologics
For thermosensitive biologics, you usually cannot rely on terminal sterilization; instead, the aseptic filling step must carry the risk burden. That is why contamination pathways at every node—air, surfaces, film, operators—become critical when comparing BFS and FFS. [woodstocksterilesolutions]
BFS Contamination Risk Profile
BFS is widely regarded as one of the lowest-risk technologies for sterile liquid products. [linkedin]
Typical contamination risk controls in BFS include: [vxpbiologics]
- Closed forming and filling chamber with HEPA-filtered air and overpressure.
- Container formed and sealed in the same machine, avoiding any open transfer.
- No glass, so no particle shedding or breakage risk.
- Ultra-short exposure time (seconds) between container formation and sealing.
For thermosensitive biologics, BFS provides a major advantage: you can often avoid high-temperature terminal sterilization because the entire process is aseptic by design, and exposure to heat is mostly confined to the container wall before filling. This is particularly relevant for proteins, monoclonal antibodies, and some vaccine antigens that degrade rapidly under heat or radiation. [susupport]
FFS Contamination Risk Profile
By contrast, FFS must manage contamination risks along a longer, more open film path. Advanced pharmaceutical FFS systems can reach high sterility levels, but typically only when combined with robust decontamination and barrier technologies. [soontact]
Common contamination controls in aseptic FFS lines include: [linkedin]
- Continuous film decontamination via H₂O₂, UV, or steam.
- RABS or isolator systems to physically separate operators from the critical zone.
- Strict environmental monitoring, including viable and non-viable particle counts along the film path.
- Careful management of sealing jaws and nozzles, which may enter and exit the critical zone.
For thermosensitive biologics, FFS can be challenging because decontamination steps (e.g., hot surfaces, reactive sterilants) must never compromise the biologic itself. You need meticulous process validation to demonstrate that residual sterilant or heat does not affect potency or stability. [susupport]
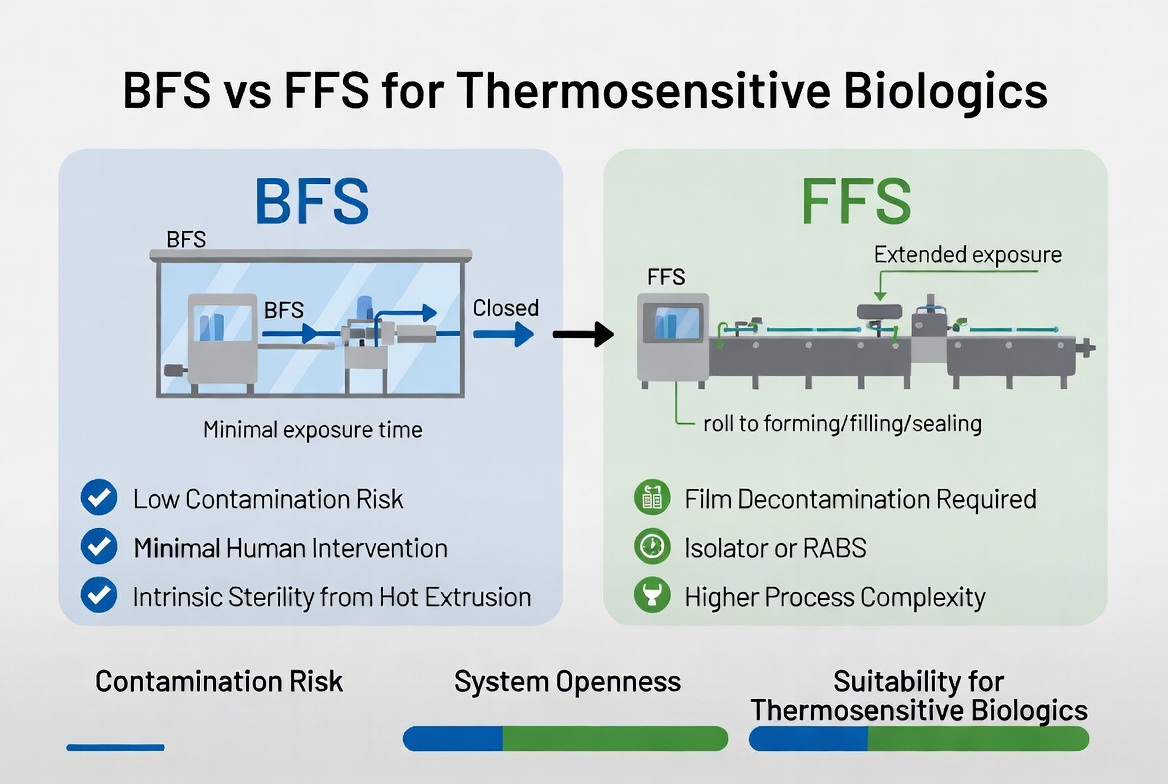
Side-by-Side Contamination & Biologic Suitability Comparison
The table below summarizes key factors that matter when your goal is to minimize contamination risk for thermosensitive biologics.
| Factor | BFS (Blow-Fill-Seal) | FFS (Form-Fill-Seal) |
| Core container material | Thermoplastic resin (LDPE, HDPE, PP) formed in-line in molten state soontact | Pre-fabricated pharmaceutical-grade films, sometimes multi-layer laminates soontact |
| System openness | Highly closed; forming, filling, sealing in one enclosed aseptic chamber soontact | More open film path; requires decontamination, airflow management, and barriers soontact |
| Human intervention | Minimal during critical steps, high automation soontact | Higher potential for intervention; mitigated by RABS/isolators and SOPs rommelag |
| Exposure time | Seconds between formation and seal soontact | Longer travel and dwell times along the line soontact |
| Typical contamination risk | Among the lowest in aseptic fill-finish, strong regulatory support soontact | Highly dependent on design and controls; risk increases with poor barrier or decontamination rommelag |
| Fit for thermosensitive biologics | Very strong, especially where terminal sterilization is not feasible vxpbiologics | Possible, but demands careful film, sterilant, and thermal impact validation susupport |
| Typical biologic formats | Ophthalmics, inhalation products, injectables, some vaccines vxpbiologics | Some biologic bags, diluents, bulk solutions in flexible packaging rommelag |
Expert Perspective – When BFS Clearly Leads for Thermosensitive Biologics
From an industry practitioner's perspective, there are clear scenarios where BFS is the default choice if you want to minimize contamination risk for thermosensitive biologics. Industry case reports indicate increasing adoption of BFS for preservative-free ophthalmic solutions, complex injectables, and inhaled biologics, precisely because these products cannot tolerate aggressive sterilization regimes. [woodstocksterilesolutions]
In my own project reviews with sterile facilities, three recurring drivers push teams toward BFS for thermolabile products:
1. Regulatory confidence in aseptic design – BFS is described in FDA and EMA guidance as an advanced aseptic process, which simplifies justification compared to highly customized FFS aseptic concepts. [apiject]
2. Reduced operator-related risk – BFS runs with very few manual interventions inside the critical zone, reducing contamination from gowning, glove integrity breaches, and human error. [linkedin]
3. Container integrity and particulate control – Uniform, in-line molded polymer containers reduce the risk of particulates from glass breakage and fragmented stoppers, issues that are particularly critical for injectables. [vxpbiologics]
For biologics with narrow stability margins, these factors often outweigh the reduced flexibility of BFS in package design. [vxpbiologics]
Where a High-Performance FFS Line Still Makes Sense
That said, FFS is not automatically disqualified for thermosensitive biologics. Several large pharma companies successfully use high-spec FFS systems to aseptically fill amino acid solutions, parenteral nutrition, and some biologic solutions into flexible bags. [rommelag]
Situations where FFS can be viable include: [linkedin]
- The biologic is moderately thermosensitive but stable within the thermal range used for film sealing and sterilant aeration.
- You need large-volume flexible bags (e.g., 250 ml–3 L) where BFS tooling would be cost-prohibitive or where rigid containers are undesirable.
- You want multi-layer films for enhanced oxygen and moisture barrier, which can extend biologic shelf life.
However, you must invest heavily in:
- Isolator/RABS technology over the entire forming, filling, and sealing path.
- Validated film decontamination solutions and continuous monitoring.
- Deep process validation to ensure no residual sterilant interacts with the biologic. [susupport]
In short, FFS can work, but it typically requires more complex engineering and validation to achieve the same level of contamination control that BFS provides more intrinsically. [soontact]
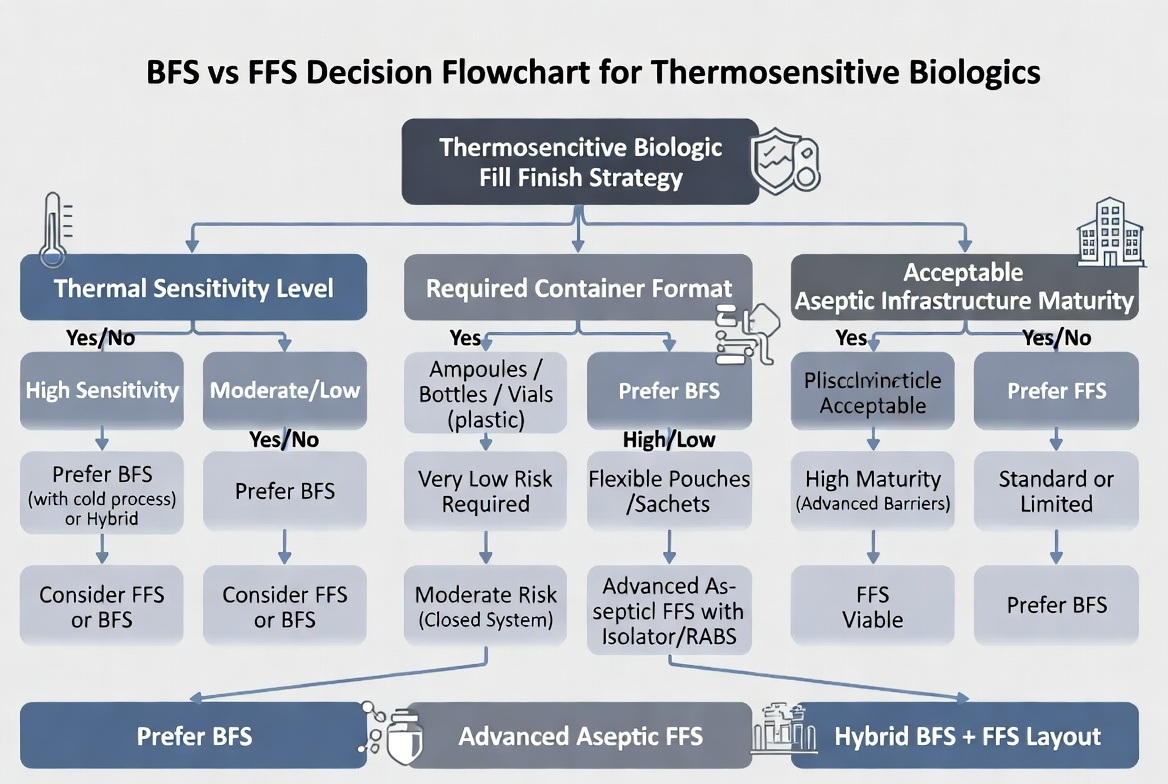
Practical Selection Framework for Biologic Manufacturers
As an equipment supplier and line integrator, Ningbo Everheal Medical Equipment Co., LTD. can help clients structure the decision around thermosensitive biologics with a simple five-question framework:
1. How Thermosensitive Is Your Biologic?
- If the product cannot tolerate terminal sterilization and is highly sensitive to heat, BFS often becomes the safer baseline option. [vxpbiologics]
- For moderately sensitive products, advanced aseptic FFS may still be considered, provided that sealing temperatures and sterilant exposure remain far from degradation thresholds. [susupport]
2. What Container Format Does Your Therapy Need?
- Single-dose ampoules, small vials, or unit-dose respiratory products align naturally with BFS. [woodstocksterilesolutions]
- Large flexible bags or multi-compartment pouches may favor FFS, as film-based systems offer more geometric flexibility. [rommelag]
3. What Contamination Risk Level Can You Accept?
- High-value biologics with low batch volume and high clinical impact per dose usually justify BFS, because the cost of a contamination event is unacceptable. [soontact]
- High-volume, lower-margin solutions may accept well-controlled FFS risk if backed by strong barrier technology and monitoring. [rommelag]
4. How Mature Is Your Aseptic Infrastructure?
- A site without isolator expertise but willing to invest in a self-contained BFS line can accelerate go-live with less complexity. [linkedin]
- Sites with experienced isolator teams and strong environmental monitoring may leverage that competence to run aseptic FFS lines efficiently. [rommelag]
5. What Is Your Scalability and Lifecycle Plan?
- BFS provides excellent scalability for high-speed, high-volume liquid biologic packaging. [apiject]
- FFS supports multi-product strategies where you may want to fill liquids, powders, and combination products on the same line over time. [linkedin]
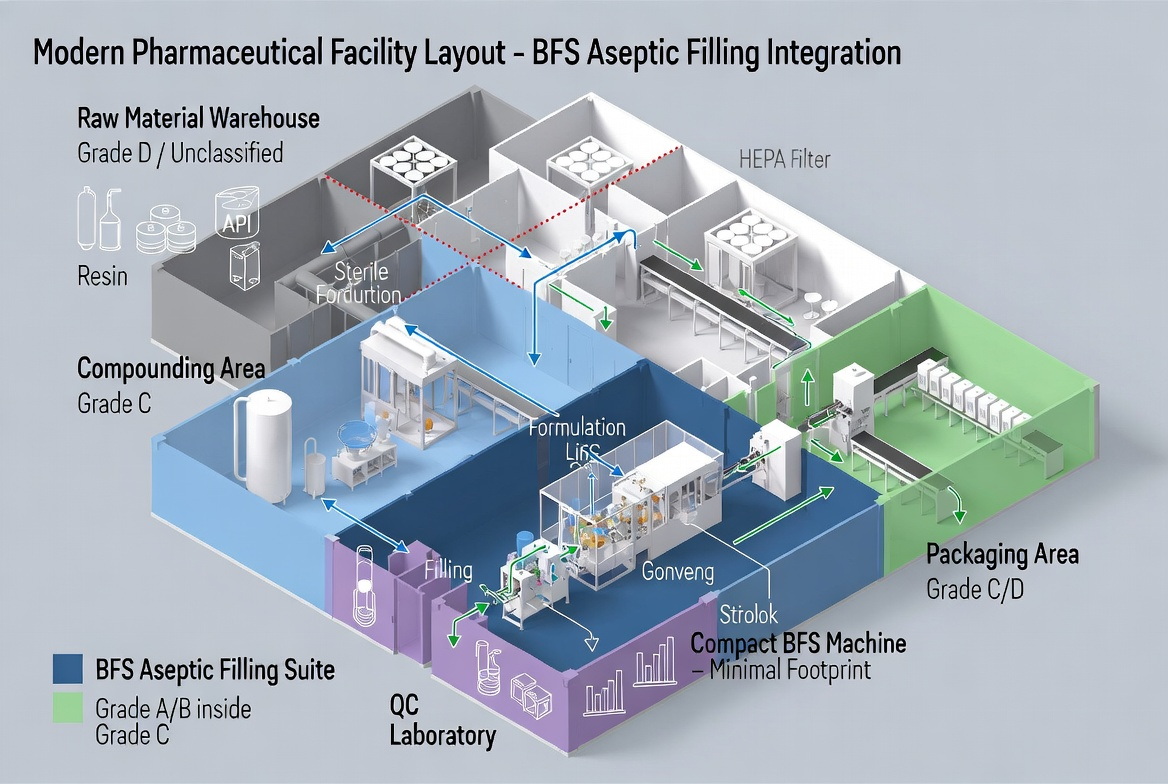
Expert Implementation Tips from a Line Engineering Perspective
Drawing on typical BFS and FFS projects delivered to pharma clients, there are several practical steps to further minimize contamination risk regardless of which technology you choose.
Engineering Tips for BFS Lines
- Design the BFS suite with unidirectional personnel and material flows, minimizing cross-contamination opportunities between clean and less-clean zones. [woodstocksterilesolutions]
- Combine BFS with automated in-line inspection (visual and leak testing) to detect microscopic seal or particle issues early. [apiject]
- Standardize on pre-qualified resins and tightly control resin handling to avoid particulate or microbial contamination before extrusion. [soontact]
Engineering Tips for FFS Lines
- Place the entire forming and filling path inside an isolator or RABS with independent pressure control, not just the filling station. [susupport]
- Integrate online film decontamination and continuously monitor sterilant concentration and exposure time. [rommelag]
- Use risk-based zoning to ensure that interventions, maintenance, and sampling never compromise the highest-grade aseptic areas. [susupport]
For Ningbo Everheal's customers, an integrated project approach—covering layout design, HVAC strategy, and line configuration—often has more impact on real-world contamination rates than the BFS vs. FFS choice alone. [woodstocksterilesolutions]
Conclusion – Which Technology Minimizes Contamination Risks for Thermosensitive Biologics?
From both literature and plant-level experience, BFS generally offers the lowest intrinsic contamination risk for thermosensitive biologics, thanks to its closed, automated, high-temperature forming process and very short exposure times. A well-engineered FFS line can achieve high aseptic performance, but it usually requires more complex barrier technology, decontamination steps, and validation effort to reach a similar risk profile. [linkedin]
For manufacturers evaluating new fill-finish capacity, the most robust strategy is to start from the biologic's thermal and contamination risk profile, then design the factory layout and BFS/FFS configuration together, rather than treating equipment as a stand-alone decision. This is exactly where a partner like Ningbo Everheal Medical Equipment Co., LTD.—combining BFS and FFS equipment expertise with factory layout planning—can reduce project risk and time-to-market. [vxpbiologics]
FAQ
1. Is BFS always better than FFS for biologics?
Not always. BFS generally offers lower contamination risk, but for certain large-volume or special package formats, an aseptic FFS line with strong barrier technology can still be the better fit. [soontact]
2. Can BFS handle highly viscous biologic formulations?
Modern BFS systems can dose a wide range of viscosities, but extreme viscosities may require specialized pumps, nozzles, and cycle time adjustments to maintain fill accuracy and sterility. [vxpbiologics]
3. Are BFS containers compatible with cold-chain storage?
Yes. Typical BFS polymers such as LDPE and PP are compatible with refrigerated conditions, and many BFS-packaged biologics are designed for cold chain distribution. Validation is still required for each specific product. [soontact]
4. How does regulatory guidance view BFS and FFS?
Regulators recognize BFS as an advanced aseptic process with a strong track record in sterile pharmaceuticals, while FFS is accepted when supported by robust validation, barrier systems, and contamination control strategies. [apiject]
5. What is the typical implementation timeline for a new BFS line?
Depending on customization and facility readiness, full implementation—from URS to PQ—often ranges from 12 to 24 months, including equipment fabrication, installation, validation, and staff training. [woodstocksterilesolutions]
References
1. Soontact. "What Is The Difference Between Blow Fill Seal (BFS) And Form Fill Seal (FFS)?" 2025. [Link]. [soontact]
2. Shraddha Shinde. "Blow Fill Seal Technology: Ensuring Sterile Compliance." LinkedIn, 2025. [Link]. [linkedin]
3. VxP Biologics. "Blow-Fill-Seal." 2018. [Link]. [vxpbiologics]
4. Rommelag. "Form Fill Seal: Aseptic Filling of Pharmaceuticals." 2026. [Link]. [rommelag]
5. ApiJect. "The Evolution and Benefits of Blow-Fill-Seal Technology in Pharma." 2024. [Link]. [apiject]
6. SUSupport. "Aseptic Filling & Processing of Biologics." 2024. [Link]. [susupport]
7. Sunil Amin. "FFS and BFS: Packaging Technologies for Pharmaceuticals." LinkedIn, 2025. [Link]. [linkedin]
8. Woodstock Sterile Solutions. "Advanced Aseptic Manufacturing Solutions." [Link]. [woodstocksterilesolutions]






















































