Choosing between rotary and linear filling and capping machines is no longer just a mechanical preference; it is a strategic decision that shapes your *speed, precision, and compliance* in small‑batch ophthalmic lines. For producers working with sterile eye drops, single-dose BFS ampoules, and small multi-dose bottles, the right configuration can mean the difference between profitable flexibility and chronic bottlenecks. [medicalexpo.com]
As a China-based pharmaceutical equipment manufacturer, Ningbo Everheal Medical Equipment Co., LTD. works daily with ophthalmic producers who struggle to balance high accuracy requirements (down to ±0.5% filling accuracy) with the need for fast changeovers and short validation cycles. This real-world experience underpins the comparison below. [everhealgroup]
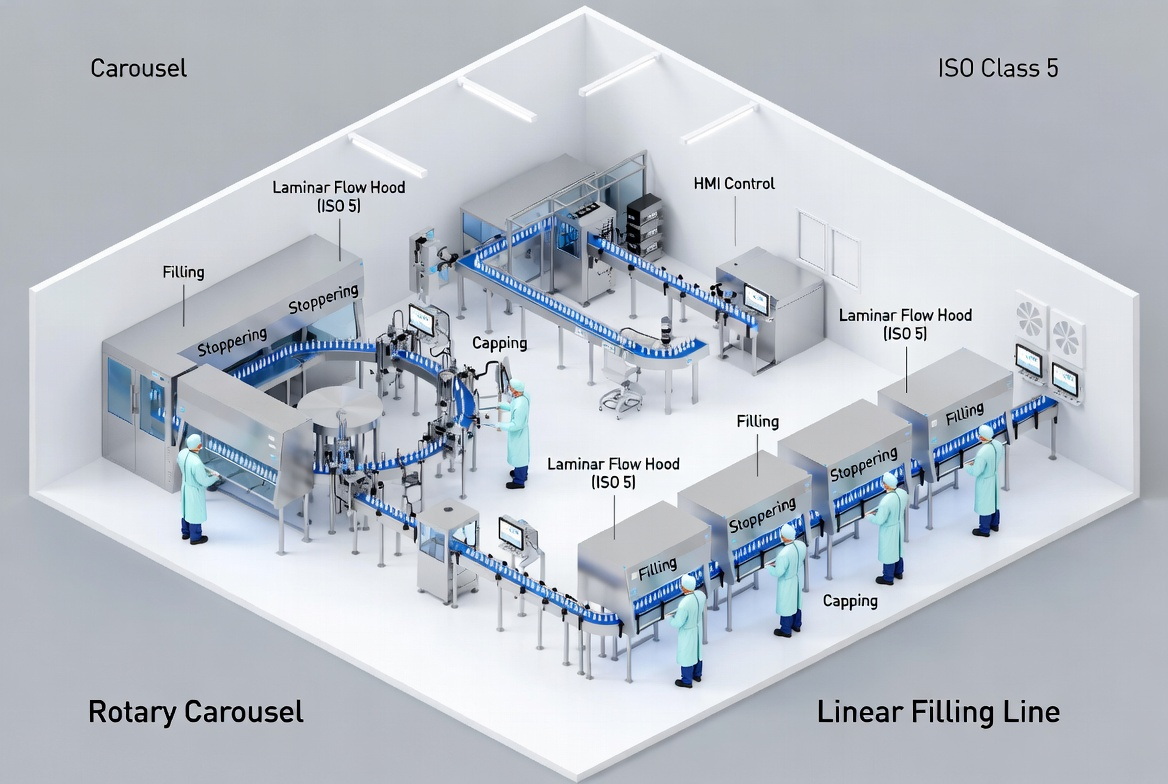
Core Definitions and Use Cases
What Is a Rotary Filling and Capping Machine?
A rotary filling and capping machine uses a circular (carousel) layout where containers travel around a central turret through each station: infeed, filling, stoppering (if required), and capping. [medicalexpo.com]
Key technical features for ophthalmic lines:
- Continuous or intermittent motion for high throughput.
- Multiple filling heads operating simultaneously on each rotation.
- Compact footprint with integrated capping and inspection options. [medicalexpo.com]
Rotary systems are widely used on high‑speed bottle‑filling lines for non‑parenteral pharmaceuticals and OTC eye drops where batch sizes are large and SKU changes are limited. [medicalexpo.com]
What Is a Linear Filling and Capping Machine?
A linear filling and capping machine moves containers in a straight path through successive stations: unscrambling (or manual feed), filling, plugging, capping, and sometimes labeling or coding. [medicalexpo.com]
Typical characteristics:
- Modular, station‑by‑station layout that is easy to reconfigure.
- Straightforward access for operators, format change, and maintenance.
- Well-suited for lower to medium speeds and frequent product changeovers. [medicalexpo.com]
For small‑batch ophthalmic fills—clinical lots, pilot runs, and multi‑SKU portfolios—linear machines often become the preferred backbone of the line.
Speed vs. Precision – The Central Trade‑Off
Speed Dynamics in Small‑Batch Ophthalmic Lines
From a throughput perspective, rotary systems typically win on pure speed. A well‑designed carousel line can manage tens of thousands of bottles per hour, especially for simple fills like buffered saline or standard eye drops. [medicalexpo.com]
However, small‑batch ophthalmic producers face three practical constraints:
- Frequent recipe and format changes (0.3 ml single-dose, 5 ml, 10 ml bottles).
- Extensive cleaning and validation in an aseptic environment.
- Limited production windows between qualification runs and QC holds. [truking]
In these conditions, the *effective* speed is often limited more by changeover and cleaning time than by nominal bottle-per-hour ratings.
Expert insight from Everheal projects:
In several BFS and FFS projects for ophthalmic and respiratory products, the net daily output improved more from reduced *setup and cleaning time* than from pushing mechanical speed. Linear designs tended to deliver better "good batches per week" even when their nameplate speed was lower. [everhealgroup]

Precision, Micro‑Dosing, and Aseptic Risks
Ophthalmic products typically require high filling accuracy and strict particulate control. Under‑fill risks dosage failure, while over‑fill wastes expensive formulations and complicates container closure integrity (CCI) testing. [truking]
- Modern BFS and high‑end filling systems can achieve ±0.5% filling accuracy for liquids and ±1.0% for suspensions. [everhealgroup]
- In‑line leak testing and particulate monitoring are increasingly standard on ophthalmic lines. [truking]
Rotary and linear platforms *can* both deliver this precision, but:
- Rotary heads are more sensitive to synchronization and wear across multiple simultaneous nozzles.
- Linear heads are easier to calibrate and validate per lane, which is attractive in small batches where each gram of API is valuable.
Detailed Comparison – Rotary vs. Linear in Ophthalmic Lines
Key Criteria Overview
Table: Rotary vs. Linear Filling and Capping for Small‑Batch Ophthalmic Lines
| Criterion | Rotary Machine | Linear Machine |
| Nominal speed | Very high; ideal for large volumes and few SKUs medicalexpo.com | Medium to high; adequate for most small‑batch needs medicalexpo.com |
| Changeover time | Longer; more format parts, turret adjustments | Shorter; modular stations, fewer critical interfaces |
| Footprint | More compact for a given speed medicalexpo.com | Usually longer line; easier segregation of stations |
| Filling precision | High, but dependent on complex synchronization medicalexpo.com | High; easier fine‑tuning per lane and recipe medicalexpo.com |
| Cleaning & SIP/CIP | More complex geometry; harder access medicalexpo.com | Better access; simpler piping and enclosures medicalexpo.com |
| Aseptic risk management | Strong if enclosed; but more moving parts in Grade A | Very strong; often easier to combine with RABS/isolators |
| Best for | High‑volume eye drops, OTC lines | Clinical, small‑batch, multi‑SKU ophthalmics |
| Investment logic | Higher initial CAPEX, lower unit cost at scale medicalexpo.com | More flexible CAPEX for evolving portfolios medicalexpo.com |
Real‑World Scenario – Configuring a Small‑Batch Ophthalmic Line
A Typical Small‑Batch Ophthalmic Use Case
Consider a manufacturer with the following profile:
- 10–15 ophthalmic SKUs per year (eye drops, lubricants, antiseptics).
- Batch sizes from 5,000 to 50,000 units each.
- Need to run validation, clinical samples, and commercial batches on the same line.
In such a case, our engineering team at Ningbo Everheal often designs hybrid lines:
- Linear filling and capping as the core platform, using servo‑driven pumps and recipe‑based changeovers.
- Integrated BFS modules for preservative‑free single‑dose units where sterility and zero human intervention are paramount. [everhealgroup]
With BFS, forming, filling, and sealing are performed in a single integrated aseptic step within an ISO Class 5 environment, achieving a Sterility Assurance Level (SAL) of 10⁻⁶. For preservative‑free ophthalmic products, this significantly reduces contamination risk versus open‑vial systems. [truking]
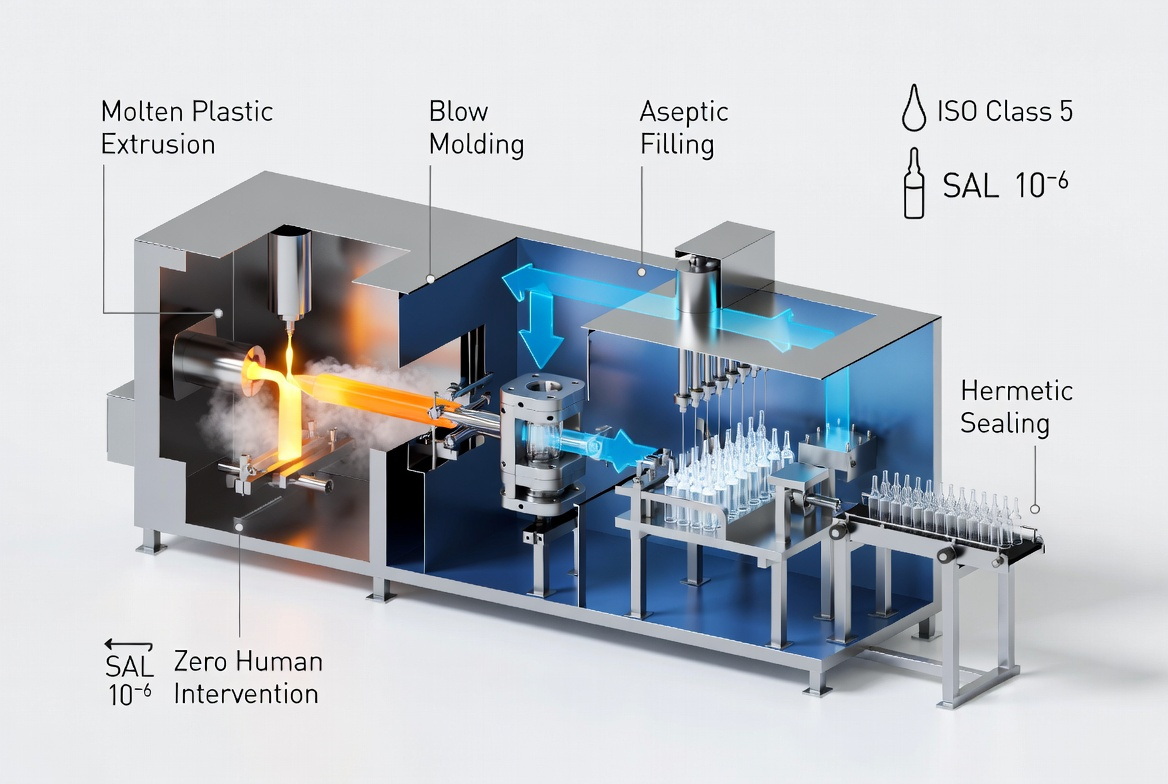
Latest Market Data and Why It Matters
Industry Growth Signals for FFS and BFS
The global market for form‑fill‑seal (FFS) machines was valued at around USD 8.7–9.2 billion in 2024–2025 and is projected to grow at approximately 5–5.2% CAGR toward 2034. This growth is driven partly by: [gminsights]
- Rising demand for pre‑sterilized, unit‑dose and convenience packaging.
- Increased regulatory scrutiny on manual interventions in sterile filling.
- The need to consolidate forming, filling, and sealing to reduce contamination risks. [mordorintelligence]
At the same time, BFS technology—already well established in ophthalmics and respiratory inhalants—continues to gain ground due to its zero human intervention in critical zones and high‑speed, container‑agnostic performance. This supports the trend toward fully enclosed, high‑precision, small‑footprint lines for sensitive liquids. [everhealgroup]
Implications for Rotary vs. Linear
As FFS and BFS adoption grows, producers reassess how rotary and linear machines fit into their overall plant layout:
- Rotary machines serve as high‑speed platforms for legacy vial and bottle formats.
- Linear lines and BFS modules become the flexible layer for new ophthalmic presentations and small‑batch developments. [truking]
From an investment standpoint, this often leads to a strategy where a linear filling and capping line is installed first, and rotary high‑speed capacity is added only after market demand stabilizes.
Expert Design Principles to Solve the Trade‑Off
Principle 1 – Start From the Product and Portfolio
From an engineering and regulatory standpoint, the correct question is not "rotary or linear?" but "what mix of products, formats, and markets will this line support over the next 5–10 years?"
Our project experience shows three practical rules:
1. If more than 60% of your annual volume is in 1–2 stable SKUs, a rotary high‑speed machine can be justified early.
2. If you handle frequent formulation and container changes, prioritize a linear configuration with recipe‑based controls and quick‑change parts.
3. If your strategic roadmap includes preservative‑free or single‑dose ophthalmics, plan from the start for BFS or FFS integration. [gminsights]
Principle 2 – Design Around Cleaning, Not Just Output
In regulated ophthalmic filling, CIP/SIP, cleaning validation, and decontamination cycles shape real productivity more than theoretical machine speed. [everhealgroup]
- Linear lines with accessible piping and enclosures reduce cleaning time and facilitate visual inspection.
- BFS systems with automated SIP and validated recipes minimize operator intervention and documentation workload. [truking]
An Everheal BFS system, for example, combines 316L stainless steel contact parts, Ra ≤ 0.25 μm surface roughness, and fully validated CIP/SIP to support rapid, repeatable cleaning that stands up to audits. [everhealgroup]
Principle 3 – Build Digital and Regulatory Readiness In
To satisfy regulators and auditors, modern lines must embed data integrity and electronic records from day one. Systems that comply with FDA 21 CFR Part 11, EU GMP Annex 1, and relevant ISO standards give you a stronger foundation for future inspections. [everhealgroup]
Both rotary and linear machines should be evaluated on:
- Availability of 21 CFR Part 11‑compliant control systems.
- In‑line inspection (leak test, fill volume, vision systems).
- Full electronic batch records and event logging. [truking]
Everheal's BFS solutions, for instance, integrate in‑line 100% leak testing, particulate monitoring, and automated reject handling, minimizing manual checks and improving data completeness. [everhealgroup]
Practical Selection Checklist for Plant Managers
Step‑by‑Step Decision Framework
Use this simple 5‑step checklist when deciding between rotary and linear systems for your small‑batch ophthalmic line:
1. Quantify batch structure
- Annual SKUs, batch size distribution, format complexity.
2. Map regulatory and sterility risks
- Aseptic class, preservative use, single‑dose vs multi‑dose.
3. Define layout constraints
- Cleanroom size, existing utilities, operator flow.
4. Score each technology
- Use criteria from the comparison table: speed, changeover, cleaning, CAPEX/OPEX.
5. Simulate future scenarios
- Add 2–3 hypothetical SKUs or regulatory changes and see which platform remains viable.
In Everheal projects, we often combine this checklist with 3D factory layout simulation to validate operator flow, equipment spacing, and material handling paths before committing to a final design. [pmecchina]
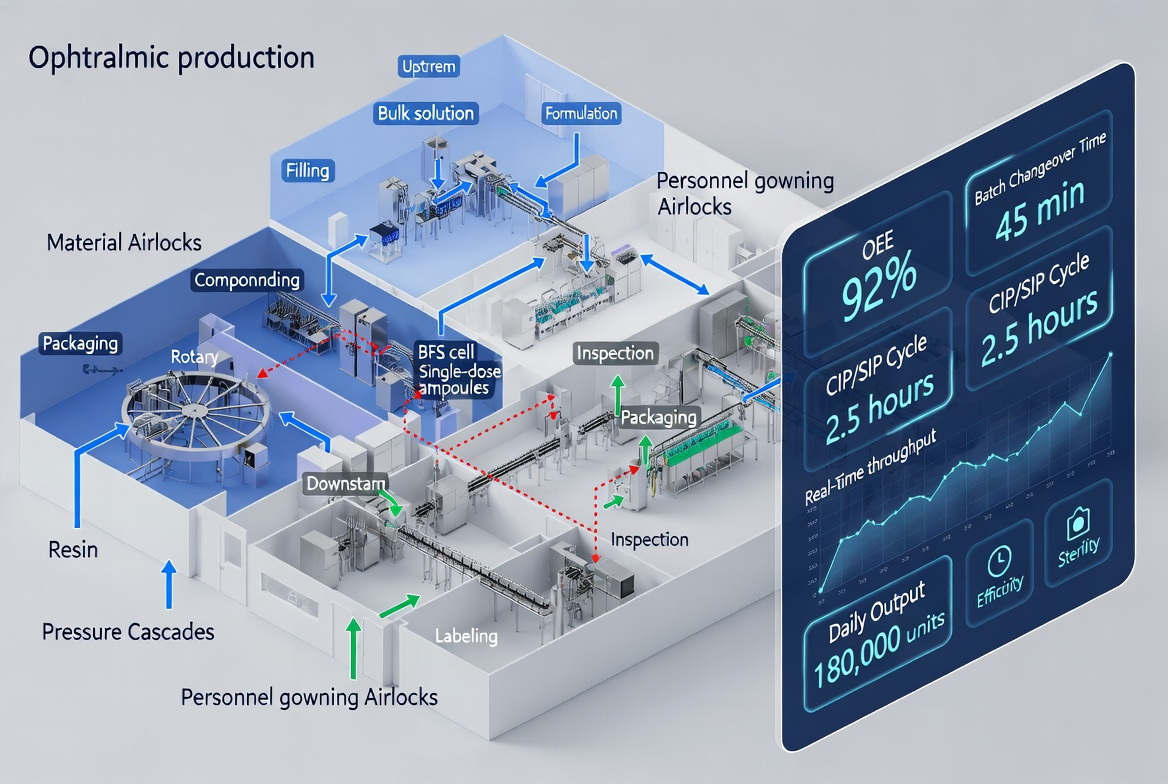
How Everheal Supports Your Ophthalmic Line Strategy
Integrated Line and Layout Engineering
As a specialist in filling and sealing machines, FFS systems, and BFS lines, Ningbo Everheal Medical Equipment Co., LTD. offers end‑to‑end engineering support for ophthalmic manufacturers: [truking]
- Custom line configuration combining rotary, linear, and BFS where appropriate.
- Factory layout planning, including cleanroom zoning, material/personnel flows, and utility mapping. [pmecchina]
- Regulatory‑aligned design, referencing ISO 13485, EU GMP Annex 1, and FDA 21 CFR 210/211. [everhealgroup]
Our BFS solutions are engineered for high‑speed production (up to 15,000 units per hour), integrated aseptic processing, and energy‑efficient servo drives that cut power consumption by about 30% compared to conventional models. [everhealgroup]
Lifecycle Service and Digital Support
Beyond installation, we provide:
- Predictive maintenance with IoT‑enabled component monitoring.
- Annual service contracts aligned with your shutdown windows.
- 24/7 global spare parts network to protect your OEE. [everhealgroup]
These services are particularly critical for ophthalmic lines, where unscheduled downtime quickly impacts clinical trial timelines and market supply.
CTA – Plan Your Next Ophthalmic Line With Confidence
If you are evaluating rotary vs. linear filling and capping machines for a new or upgraded ophthalmic line, the safest approach is to build your decision on *data, portfolio analysis, and layout simulations*—not just nameplate speed.
Talk to the Everheal engineering team to model your batches, design a compliant layout, and configure the optimal combination of linear, rotary, and BFS/FFS technologies for your small‑batch ophthalmic production.
FAQs: Rotary vs. Linear Filling and Capping in Ophthalmics
Q1: Are rotary machines always better for ophthalmic production because they are faster?
A1: Not necessarily. While rotary machines often deliver higher nominal speeds, small‑batch ophthalmic lines are limited by cleaning, changeover, and validation time, where linear machines may outperform in real effective output. [medicalexpo.com]
Q2: Can linear machines achieve the same filling accuracy as rotary machines?
A2: Yes. Modern linear systems with servo‑driven pumps can reach filling accuracies around ±0.5% for liquids, comparable to high‑end rotary lines, and they are often easier to calibrate per lane for different recipes. [medicalexpo.com]
Q3: When should I consider BFS instead of conventional bottle filling?
A3: BFS is particularly attractive for preservative‑free eye drops and single‑dose units, where integrated forming, filling, and sealing in an ISO Class 5 environment and SAL 10⁻⁶ significantly reduce contamination risk and manual interventions. [truking]
Q4: How do regulatory expectations influence the choice between rotary and linear?
A4: Regulators focus on aseptic risk, data integrity, and cleaning validation rather than machine type. Systems that support 21 CFR Part 11, Annex 1 compliance, robust CIP/SIP, and in‑line inspection will be favored regardless of rotary or linear design. [pmecchina]
Q5: What is the first step if I want to upgrade my existing ophthalmic line?
A5: Start by mapping your current SKUs, batch sizes, downtime drivers, and regulatory gaps, then work with an equipment partner like Everheal to simulate different configurations (linear, rotary, BFS/FFS) and quantify the impact on throughput and compliance. [mordorintelligence]
References
1. MedicalExpo – Pharmaceutical filling and sealing machines overview. Available at: [https://www.medicalexpo.com.cn/zhizaoshang-yiliao/zhi-yao-guan-zhuang-feng-kou-ji-50863.html] [medicalexpo.com]
2. MedicalExpo – Aseptic filling and sealing machines. Available at: [https://www.medicalexpo.com.cn/zhizaoshang-yiliao/wu-jun-guan-zhuang-feng-kou-ji-50905.html] [medicalexpo.com]
3. Global Market Insights – Form‑Fill‑Seal (FFS) machine market data 2024–2034. Available at: [https://www.gminsights.com/zh/industry-analysis/form-fill-seal-machines-market] [gminsights]
4. Mordor Intelligence – Form‑Fill‑Seal packaging machine market outlook. Available at: [https://www.mordorintelligence.com/zh-CN/industry-reports/form-fill-seal-packaging-machine-market] [mordorintelligence]
5. Everheal Medical Equipment Co., LTD. – BFS Machine Manufacturer technical and compliance information. Available at: [https://www.everhealgroup.com/bfs-machine-manufacturer.html] [everhealgroup]
6. PMEC China – Intelligent pharmaceutical packaging and smart plant trends. Available at: [https://www.pmecchina.com/archives/58613] [pmecchina]
7. Truking – Blow‑Fill‑Seal solution overview (PDF). Available at: [https://www.truking.com/uploadfiles/2024/09/20240909141902002.pdf] [truking]






















































