In modern injectable and biologics facilities, pyrogen‑free glassware is non‑negotiable for patient safety and regulatory compliance. Pharmaceutical manufacturers now face a strategic choice: continue relying on moist heat autoclaves for "all‑purpose" sterilization, or deploy dedicated dry heat sterilization cabinets optimized for high‑throughput depyrogenation of vials, ampoules, and other primary packaging. Drawing on field experience supporting global sterile filling plants, Ningbo Everheal Medical Equipment Co., LTD designs and manufactures pharmaceutical sterilization cabinets and related production line equipment that sit at the heart of these decisions. [everhealgroup]

Fundamentals – How Dry Heat and Moist Heat Sterilization Work
Dry Heat Sterilization Cabinets – High‑Temperature Oxidative Depyrogenation
Dry heat sterilization cabinets use circulated hot air at very high temperatures, typically in the 160–300 °C range, to sterilize and depyrogenate glassware. At these temperatures, microorganisms and endotoxins are destroyed primarily via oxidative processes that denature critical biomolecules and cell structures. For depyrogenation, temperatures around 250 °C held for at least 1 hour are commonly cited in SOPs and industry practice to achieve validated endotoxin reduction (often ≥3 log reduction in endotoxin challenge tests). [princesterilization]
In practice, modern dry heat sterilization cabinets for glassware offer:
- Forced hot‑air circulation for uniform temperature distribution across densely loaded vial or ampoule racks. [zhishi.shujunyiqi]
- HEPA‑filtered airflow to maintain a controlled clean environment within the chamber. [zhishi.shujunyiqi]
- Precision control to meet validated time–temperature profiles for depyrogenation cycles. [pharmastate]
Because no water or steam is involved, dry heat is especially suitable for high‑temperature‑resistant materials such as borosilicate glass, metal caps, and certain aluminum components. [thepharmavision]
Moist Heat Autoclaves – Steam, Pressure, and Protein Denaturation
Moist heat sterilization uses saturated steam under pressure to rapidly transfer heat and denature microbial proteins. This is typically achieved in an autoclave operating at 121 °C for around 15–30 minutes (at approximately 1 bar overpressure) to achieve the required sterility assurance level. Compared with dry heat, moist heat sterilization is more efficient at killing microorganisms at lower temperatures and shorter exposure times. [gruenberg]
Key characteristics of moist heat autoclaves include:
- Use of steam penetration and latent heat to destroy microbes.
- Shorter cycles and lower temperatures than dry heat for pure sterilization purposes. [duralinesystems]
- Broad applicability for liquids, textiles, rubber, and plastic components that cannot tolerate dry heat at 250 °C. [duralinesystems]
However, while autoclaves provide excellent sterilization, they are less effective for depyrogenation, because endotoxins require significantly higher temperatures for reliable inactivation. [pharmastate]
Depyrogenation Requirements for Pharmaceutical Glassware
What Depyrogenation Really Means in Practice
Depyrogenation is not just "extra sterilization." It is the validated reduction of pyrogens—primarily bacterial endotoxins—to below defined specification limits on product‑contact surfaces. Industry SOPs for injectable production often recommend depyrogenating cleaned glassware in hot air ovens at around 250 °C for not less than 1 hour to ensure sufficient removal or inactivation of endotoxins. [thepharmavision]
In a typical sterile injectables facility:
- Empty glass vials, ampoules, and tubes are washed, dried, and then depyrogenated by dry heat before entering aseptic filling. [zhishi.shujunyiqi]
- Components made from rubber, silicon, or polyethylene are sterilized in autoclaves rather than exposed to depyrogenation temperatures. [thepharmavision]
This division of roles reflects a practical reality: depyrogenation is a high‑temperature specialty step suited to dry heat, not a simple extension of moist heat sterilization. [pharmastate]
Why Dry Heat Is the Gold Standard for Glassware Depyrogenation
From a process engineer's standpoint, dry heat sterilization cabinets offer several structural advantages for depyrogenating glassware:
- Higher achievable temperatures (e.g., 250 °C) compared with autoclaves, which typically operate around 121–134 °C. [princesterilization]
- Proven depyrogenation SOPs recommending dry heat ovens for glassware used in endotoxin‑sensitive tests and injectable production. [pharmastate]
- Ability to handle tightly packed glassware in metal or stainless‑steel trays without concerns about steam penetration or condensate. [zhishi.shujunyiqi]
Industry guidelines and real‑world SOPs consistently treat dry heat ovens and depyrogenation tunnels as the primary solution for removing endotoxins from glassware, while reserving autoclaves for sterilizing garments, rubber stoppers, filters, and plastic accessories. [thepharmavision]

Side‑by‑Side Comparison – Dry Heat Sterilization Cabinets Vs. Moist Heat Autoclaves
Key Technical and Operational Differences
Below is a practical, high‑level comparison for engineering and QA teams deciding how to handle glassware depyrogenation.
| Aspect | Dry Heat Sterilization Cabinets | Moist Heat Autoclaves |
| Core principle | Hot air at high temperature causing oxidative destruction of microbes and endotoxins (princesterilization) | Saturated steam under pressure causing protein denaturation of microbes (princesterilization) |
| Typical conditions | 160–180 °C for sterilization; around 250 °C for ≥1 h for depyrogenation (zhishi.shujunyiqi) | 121 °C for 15–30 minutes for sterilization (princesterilization) |
| Depyrogenation suitability | Highly suitable for glassware; standard method in SOPs (zhishi.shujunyiqi) | Limited; temperature often insufficient for endotoxin inactivation (thepharmavision) |
| Compatible loads | Glass vials, ampoules, metal components, some aluminum parts (zhishi.shujunyiqi) | Liquids, rubber stoppers, garments, plastic components, filters (thepharmavision) |
| Energy and utilities | Electric or gas heating; no steam network required (zhishi.shujunyiqi) | Requires high‑quality steam, condensate management, and water (princesterilization) |
| Cycle time | Longer heat‑up and cool‑down for depyrogenation loads (duralinesystems) | Shorter cycles at lower temperatures for sterilization only (princesterilization) |
| Maintenance | Less piping, valves, and seals than steam systems; lower maintenance burden (gruenberg) | More complex steam, vacuum, and condensate systems; higher maintenance needs (gruenberg) |
| Impact on materials | Suitable only for high‑temperature‑resistant materials (zhishi.shujunyiqi) | Suitable for many heat‑sensitive materials that cannot withstand 250 °C (thepharmavision) |
For injectable glassware, this table makes one fact clear: dry heat sterilization cabinets are the preferred platform when depyrogenation, not just sterilization, is the critical requirement. [pharmastate]
Expert Perspective – How Real Plants Combine Both Technologies
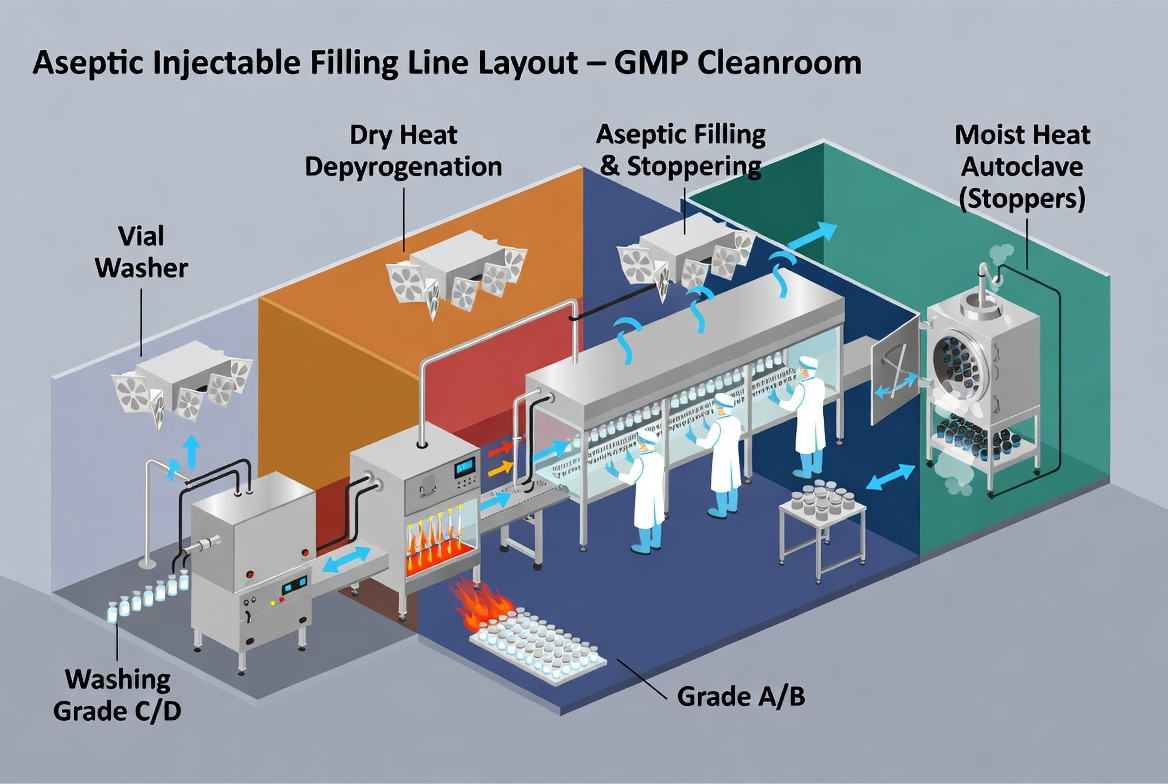
Typical Layout in a Modern Sterile Filling Line
In practice, top‑performing sterile facilities do not choose between dry heat and moist heat; they integrate both technologies in a complementary way. A conventional layout for freeze‑dried or liquid injectables may look like this: [linkedin]
1. Component preparation
- Glass vials and ampoules: Washed, then sent through a dry heat depyrogenation cabinet or tunnel at around 250 °C. [zhishi.shujunyiqi]
- Rubber stoppers, gaskets, and tubing: Washed and sterilized in an autoclave at around 121–124 °C. [thepharmavision]
2. Aseptic filling and stoppering
- Depyrogenated vials enter a Grade A environment for filling and partial stoppering. [linkedin]
3. Lyophilization or terminal processing
- Vials may be transported directly to a freeze‑dryer or to further capping and inspection equipment. [linkedin]
From my experience supporting clients scaling from local to global markets, the most robust designs use dry heat for depyrogenating glassware and autoclaves for ancillary loads, avoiding the temptation to push autoclaves into roles they are not optimized for. This separation of duties simplifies validation and supports clearer lifecycle documentation.
Case‑Style Insight – Endotoxin‑Sensitive Workflows
Industry SOPs for endotoxin‑sensitive analysis and BET sampling explicitly call for depyrogenating glassware in hot air ovens, while using autoclaves for plastic and rubber apparatus. For example: [thepharmavision]
- All empty glassware and accessories used for bacterial endotoxin testing are preferably sterilized and depyrogenated in a hot air oven. [thepharmavision]
- Apparatus made of rubber, silicon, or polyethylene are instead sterilized in the autoclave. [thepharmavision]
This dual strategy mirrors what we see in high‑throughput injectable lines: dry heat is the tool of choice when endotoxins are the limiting factor, while autoclaves remain indispensable for mixed‑material components and media.
Validation, Compliance, and Risk Management
Meeting Depyrogenation and Sterility Requirements
Regulators and auditors expect clear, validated time–temperature profiles for both sterilization and depyrogenation steps. For depyrogenation in dry heat ovens, SOPs commonly require exposure of cleaned glassware at 250 °C for a minimum of 1 hour, with documented evidence of temperature uniformity and load mapping. [pharmastate]
For autoclaves, validation focuses on:
- Demonstrating steam penetration into representative loads.
- Monitoring temperature and pressure profiles to meet target F₀ or cycle parameters. [princesterilization]
- Maintaining detailed batch records that track each load, including temperature and pressure data. [thepharmavision]
Well‑designed pharmaceutical sterilization cabinets and autoclaves support this by integrating data recording and batch reporting, making it easier for QA to review logs and for engineering to troubleshoot deviations. [everhealgroup]
Risk‑Based Technology Selection
From a risk management standpoint, the question to ask is: "What is the worst‑case impact if depyrogenation is incomplete?" For injectable glassware and endotoxin‑sensitive tests, the answer is patient harm or failed batches, which justifies dedicated dry heat depyrogenation systems. [pharmastate]
Autoclaves are still critical for:
- Sterilizing garments, accessories, and reusable equipment. [thepharmavision]
- Handling mixed loads containing heat‑sensitive polymers and elastomers.
In other words, you mitigate risk by assigning each technology to the load types it handles best, and by not expecting an autoclave to do the work of a depyrogenation oven.
Operational and Economic Considerations for Plant Managers
Efficiency, Maintenance, and Utility Load
When we model total cost of ownership across a 10‑year equipment lifecycle, the differences between dry heat cabinets and autoclaves become clearer:
- Dry heat sterilization cabinets
- No steam generation system, reducing complexity and utilities. [gruenberg]
- Simpler piping and fewer critical seals, which can mean less maintenance time compared with steam autoclaves. [gruenberg]
- Longer cycles for depyrogenation, but potentially offset by high‑capacity racks and continuous or batch operation. [duralinesystems]
- Moist heat autoclaves
- Require continuous access to high‑quality purified water and steam. [princesterilization]
- More intensive maintenance on piping, valves, gaskets, and condensate systems. [gruenberg]
- Fast cycles, ideal for high‑frequency sterilization of small equipment batches. [duralinesystems]
For glassware depyrogenation, many facilities find that dedicated dry heat capacity reduces bottlenecks by removing glass loads from multi‑purpose autoclaves, freeing those systems for other critical tasks.
Layout and Integration with Upstream/Downstream Equipment
As a manufacturer of pharmaceutical sterilization cabinets and turnkey layout solutions, Ningbo Everheal can help clients integrate dry heat depyrogenation directly into vial washing and filling lines. Common integration points include: [everhealgroup]
- In‑line transfer of washed vials from washer outlet to dry heat depyrogenation cabinet.
- Automated unloading from the cabinet into Grade A filling environments.
- Synchronization of cabinet cycle times with upstream washing and downstream filling speeds.
Practical Selection Framework – Which Method Should You Use for Glassware Depyrogenation?
Step‑by‑Step Decision Guide
If you are evaluating options for a new line or upgrading an existing plant, you can use the following simplified framework:
1. Define your critical loads
- Are you primarily handling glass vials, ampoules, and tubes for injectables or endotoxin‑sensitive tests? If yes, prioritize dedicated dry heat depyrogenation capacity. [zhishi.shujunyiqi]
2. Assess endotoxin requirements
- If your process requires validated depyrogenation (e.g., BET‑related glassware, injectable containers), align your URS with 250 °C for ≥1 hour dry heat capability. [pharmastate]
3. Map material constraints
- Separate glass and metal components (dry heat) from rubber, plastic, and textile components (autoclave). [thepharmavision]
4. Evaluate utilities and maintenance
- If steam capacity is limited or expensive, using dry heat cabinets for high‑temperature glassware loads can offload demand from autoclaves and simplify utility planning. [gruenberg]
5. Plan validation and documentation
- Ensure both dry heat and moist heat equipment provide traceable records, load mapping, and support for your QA documentation workflows. [pharmastate]
Following this kind of structured decision process usually leads to a hybrid configuration where dry heat sterilization cabinets handle glassware depyrogenation and autoclaves focus on diversified sterilization tasks.
How Ningbo Everheal Supports Your Depyrogenation Strategy
As a specialized manufacturer of pharmaceutical sterilization cabinets and production line solutions, Ningbo Everheal Medical Equipment Co., LTD helps global clients design systems that balance sterilization, depyrogenation, and throughput. With experience in layout planning and customized equipment configurations, Everheal can provide: [everhealgroup]
- Dry heat sterilization cabinets tailored for vial and ampoule depyrogenation, meeting validated time–temperature profiles. [everhealgroup]
- Integration with upstream washing and downstream filling, freeze‑drying, and capping equipment for a seamless line concept. [linkedin]
- Engineering support to coordinate dry heat and moist heat capacity, ensuring your autoclaves and sterilization cabinets are each used where they add the most value. [everhealgroup]
If you are planning a new injectable plant or upgrading existing capacity, engaging early with equipment and layout specialists reduces both technical and regulatory risk.
Conclusion & Call to Action
For depyrogenation of glassware, dry heat sterilization cabinets clearly provide the most robust and widely validated approach, operating at temperatures that effectively inactivate endotoxins while integrating smoothly into modern sterile filling lines. Moist heat autoclaves remain indispensable for sterilizing liquids, garments, rubber stoppers, and mixed‑material loads, but they are not a substitute for dedicated dry heat depyrogenation. [princesterilization]
If you are currently debating how to balance dry heat sterilization cabinets and autoclaves in your next project, reach out to Ningbo Everheal Medical Equipment Co., LTD to discuss a tailored depyrogenation and sterilization concept for your plant, from equipment selection to full layout planning. A brief technical consultation can clarify capacity needs, validation strategy, and integration options before you commit CAPEX. [linkedin]
FAQ
1. Can a moist heat autoclave be used for depyrogenation of glassware?
Autoclaves are excellent for sterilization but typically operate at temperatures (around 121 °C) that are not sufficient for validated endotoxin inactivation on glassware, so they are not the preferred solution for depyrogenation. [princesterilization]
2. What time–temperature profile is commonly used for depyrogenating glassware in dry heat cabinets?
Industry SOPs often specify 250 °C for at least 1 hour for depyrogenation of cleaned glassware, with validated temperature mapping and documentation. Actual parameters must be justified and validated for each specific application. [pharmastate]
3. Which components should go to dry heat cabinets and which to autoclaves?
In many facilities, glass vials, ampoules, and other high‑temperature‑resistant components go to dry heat depyrogenation cabinets, while rubber stoppers, textiles, and plastic components are sterilized in autoclaves at lower temperatures. [zhishi.shujunyiqi]
4. How does using a dedicated dry heat sterilization cabinet affect overall line efficiency?
Dedicated dry heat capacity for glassware often removes bottlenecks from autoclaves, allowing them to focus on other loads; with appropriate rack design and cycle planning, depyrogenation cabinets support stable high‑throughput operation. [gruenberg]
5. Why should we involve equipment manufacturers early in plant design?
Early collaboration with specialists such as Ningbo Everheal helps align equipment sizing, utilities, validation, and layout, reducing redesign risk and avoiding hidden capacity constraints later in the project. [linkedin]
References
1. "Difference Between Moist Heat Sterilization & Dry Heat Sterilization." Prince Sterilization. [princesterilization]
2. "Comprehensive Sterilization & Depyrogenation Solutions." C & G Containers, Inc. [cgcontainersinc]
3. "Pharmaceutical Sterilization Cabinet – EVERHEAL." Everheal Group official website. [everhealgroup]
4.Shujunyiqi technical article on dry heat sterilization cabinets. [zhishi.shujunyiqi]
5. "Dry Heat vs Autoclaves." Gruenberg blog on benefits of dry heat sterilization over autoclaves. [gruenberg]
6. "SOP for Cleaning and Sterilization / Depyrogenation of Glassware & Accessories." The Pharma Vision. [thepharmavision]
7. Ningbo Everheal Medical Equipment Co., LTD updates and process insights (LinkedIn activity). [linkedin]
8. "Dry Heat Sterilizer vs Steam Sterilizer Autoclave." Duraline Systems. [duralinesystems]
9. "SOP for Depyrogenation." PharmaState Academy. [pharmastate]





















































