In high‑dust pharmaceutical labs and solution preparation areas, air filtration is not just an HVAC detail – it is a GMP‑critical control that affects sterility assurance, operator safety, and environmental footprint. As a process engineer and content strategist working with sterile manufacturing lines, I have seen how the choice between washable pre‑filters and disposable synthetic media quietly shapes energy use, filter spend, and cleanroom uptime over years of operation. This article compares both options from a practical, expert perspective, with a focus on sustainable strategies for large‑volume injection, lyophilized, and anticancer drug production environments. [erlab]
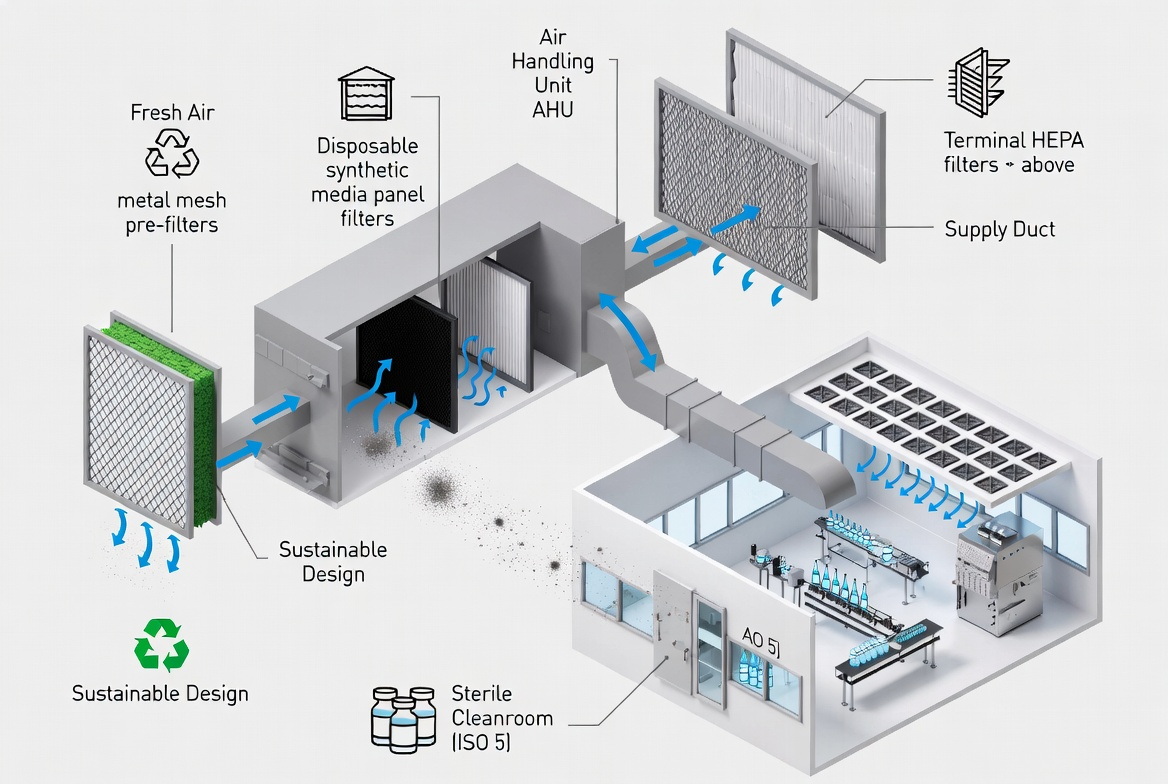
What Are Washable Pre-Filters and Disposable Synthetic Media?
Washable Pre-Filters in High-Dust Labs
Washable pre‑filters are reusable filter elements, often metal mesh or synthetic media in a rigid frame, designed to capture coarse dust and fibers before the air reaches fine or HEPA filters. In pharma HVAC systems, they typically sit at the intake or upstream of terminal HEPA filters to extend downstream filter life and stabilize pressure drop. These pre‑filters can be removed, washed (often with water and detergent), dried, and reinstalled for multiple service cycles. [cleanroomworld]
For high‑dust support areas around solution preparation tanks, utilities, and secondary packaging, washable pre‑filters provide a robust first barrier without generating large volumes of solid waste. When integrated into a validated cleaning SOP, they can significantly reduce the number of HEPA or fine filters that must be replaced each year. [directindustry]
Disposable Synthetic Media Filters
Disposable synthetic media filters use non‑woven polyester or similar synthetic fibers mounted in a cardboard or metal frame. They are engineered for one‑time use: once the dust loading or pressure drop limit is reached, the entire filter is removed and discarded. [purolatorairfilters]
These filters are widely used in commercial and light industrial HVAC because they offer predictable performance, low unit cost, and easy replacement. In pharma labs, disposable synthetic pre‑filters often sit just upstream of HEPA filters in higher‑grade rooms, where consistent filtration efficiency and clean changeout procedures are more important than reusability. [ph.parker]
Filtration Efficiency and Performance Under High Dust Load
Particle Size Capture and Lab Air Quality
Both washable pre‑filters and disposable synthetic media target larger particles than HEPA filters, but their efficiency curves look different. Washable units (especially metal mesh) are optimized for coarse dust and lint; their performance tends to drop sharply for finer particles unless engineered multi‑layer designs are used. [erlab]
Disposable synthetic media are typically designed with graded density to capture a wider band of particle sizes more efficiently at the same pressure drop. In high‑dust labs handling powders, excipients, and packaging debris, this can result in more stable indoor air quality and less migration of mid‑size particles to downstream filters. [cleanroomworld]
Pressure Drop Stability and Energy Use
Pressure drop directly affects fan energy and airflow stability in cleanrooms. Washable pre‑filters can maintain a relatively low pressure drop when properly cleaned, but performance becomes operator‑dependent: delayed washing or improper drying can create variability and even microbial risk. [sciencedirect]
Disposable synthetic media, by contrast, show a well‑characterized pressure drop curve from new to end of life, which allows engineering teams to set precise changeout criteria and balance energy versus replacement cost. For Grade B/A zones or laminar flow areas above filling machines and lyophilization lines, this predictability is often a strong operational advantage. [purolatorairfilters]
Sustainability and Lifecycle Environmental Impact
Waste Generation and Material Footprint
From a sustainability perspective, solid waste is the most visible difference. Washable pre‑filters can be reused dozens of times, drastically cutting the number of filter frames sent to landfill or incineration per year. For a large high‑dust lab with frequent pre‑filter changes, this may reduce annual filter waste mass by a significant margin. [directindustry]
Disposable synthetic media, especially in panel form, can accumulate quickly as hazardous or regulated waste if loaded with pharmaceutical dusts. However, they avoid the water and detergent footprint associated with cleaning washable units, and some manufacturers are exploring recyclable frames and lower‑impact synthetics. [sciencedirect]
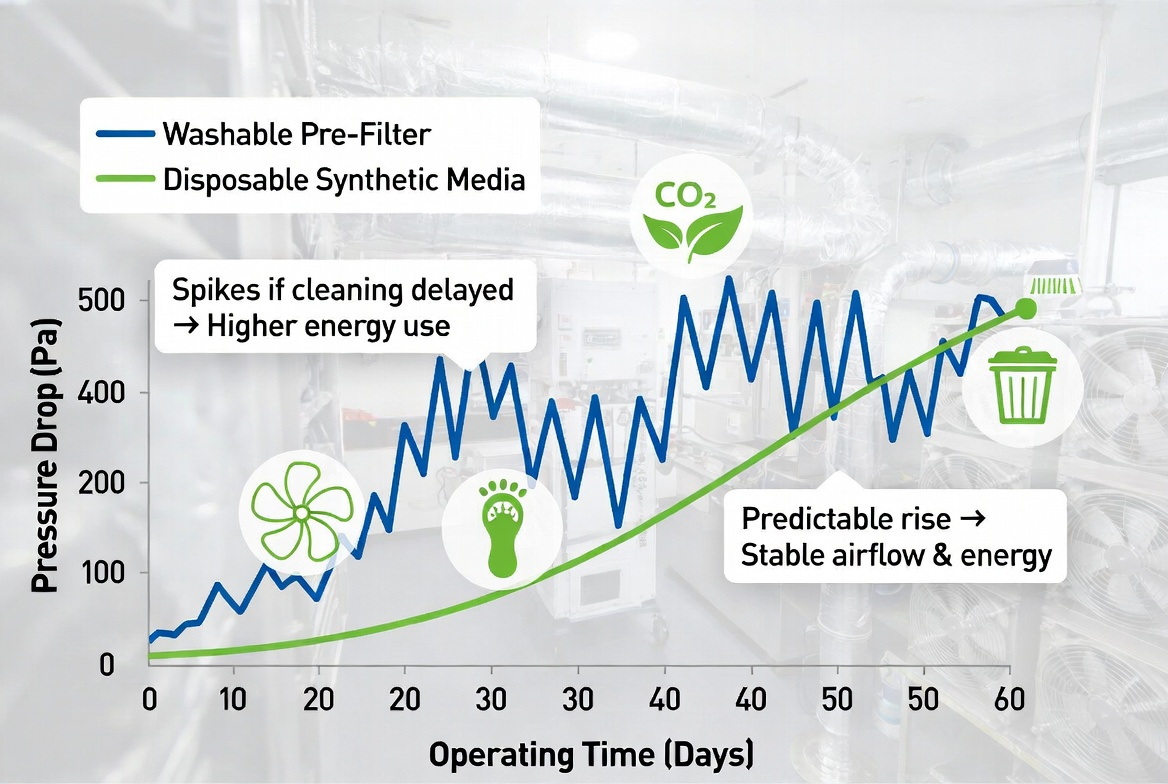
Energy Efficiency and Carbon Footprint
Air filtration is also a major energy driver for labs, which already consume far more energy per square meter than typical commercial buildings. If washable pre‑filters are not cleaned on schedule, they can create higher pressure drops that force fans to work harder, offsetting some of the environmental benefit of reuse. [mygreenlab]
Optimized synthetic media designs can offer a better efficiency‑to‑pressure‑drop ratio, so when used and changed out according to data‑driven thresholds, they contribute to lower fan energy and reduced CO₂ emissions over the system's lifecycle. A sustainable strategy in practice often combines both types: washable units in high‑dust but lower‑risk zones, and high‑performance disposables where indoor air quality and precision airflow are critical. [erlab]
Cost of Ownership in Pharma and High-Dust Labs
Direct Costs: Purchase, Cleaning, Replacement
Upfront, washable pre‑filters cost more per unit, but their cost per operating hour is often lower once multiple cleaning cycles are accounted for. The trade‑off is labor: technicians need time to remove, clean, dry, and reinstall filters, plus utilities cost for water and detergents. [directindustry]
Disposable synthetic media are cheaper to buy but must be purchased and stocked in larger quantities. In high‑dust labs with aggressive changeout intervals, these recurring costs can exceed the total cost of washable solutions within the first few years, particularly for large installations feeding multiple cleanrooms or sterile manufacturing suites. [ph.parker]
Indirect Costs: Downtime and HEPA Protection
From a pharma facility owner's perspective, the hidden cost is often downstream HEPA replacement and unplanned downtime. Inadequate pre‑filtration – whether washable or disposable – shortens HEPA life and forces expensive, disruptive shutdowns for changeout and re‑qualification. [cleanroomworld]
Well‑maintained washable pre‑filters can significantly slow HEPA loading, but the results depend on a disciplined cleaning SOP and documentation. Disposable synthetic media offer more consistent performance with less operational variability, which can be valuable where HEPA banks protect Grade B/A environments above sterilization cabinets, lyophilizers, and aseptic filling lines. [erlab]
Risk, Compliance, and GMP Considerations
Cleaning Validation and Cross-Contamination Risk
For GMP‑regulated pharma facilities, washable pre‑filters introduce a cleaning validation question: how do you prove that washing removes residual active, endotoxins, or bioburden to an acceptable level? Washing and reuse may be acceptable in non‑critical HVAC paths or technical corridors, but regulators will expect strong risk assessments and SOPs if washable filters are used near classified areas. [erlab]
Disposable synthetic media reduce this complexity: each changeout removes the captured dust and fibers permanently from the air path, lowering the risk of cross‑contamination between product campaigns. For multi‑product plants handling cytotoxics or potent APIs, this risk reduction is often decisive. [purolatorairfilters]
Documentation and Audit Readiness
Auditors look for clear, traceable maintenance records that link filtration performance with HVAC integrity and product protection. Washable pre‑filters require documented washing procedures, drying controls, inspection records, and acceptance criteria for reuse. [erlab]
Disposable synthetic media simplify documentation to changeout frequency, pressure‑drop monitoring, visual inspection, and supplier qualification. In practice, many pharmaceutical companies adopt a hybrid model: washable filters in utility and non‑classified zones, disposables in air streams feeding critical rooms, supported by a unified digital maintenance record. [ph.parker]
Expert Strategy: Hybrid Filtration Design for High-Dust Pharma Labs
Drawing on project experience with sterile solution preparation and lyophilization lines, a sustainable, GMP‑aligned approach often looks like this. [cleanroomworld]
Recommended Layered Strategy
- Use washable pre‑filters at the AHU intake or in technical zones with high dust but low GMP risk (e.g., bulk powder receiving, secondary packaging corridors). [directindustry]
- Deploy disposable synthetic media as intermediate pre‑filters upstream of HEPA filters in Grade C/B rooms and lab spaces where particle control is critical. [purolatorairfilters]
- Maintain high‑efficiency HEPA filters at terminal diffusers and critical equipment (isolators, RABS, lyophilizers, sterilization cabinets). [cleanroomworld]
- Monitor pressure drop across each stage and adjust changeout or washing frequency based on data rather than fixed calendars. [sciencedirect]
From a sustainability angle, this layered approach maximizes the reusable share of filters while preserving rigorous control where it matters most for product quality and patient safety. [sciencedirect]

Side-by-Side Comparison Table
| Factor | Washable Pre-Filters | Disposable Synthetic Media |
| Typical construction | Metal mesh or washable synthetic media in rigid frame directindustry | Non‑woven synthetic fibers in disposable panel frame ph.parker |
| Reuse potential | High; multiple wash cycles possible directindustry | None; single use, then discard ph.parker |
| Best particle range | Coarse dust and fibers directindustry | Coarse to medium particles with graded density ph.parker |
| Waste generation | Low solid waste, more wastewater from cleaning erlab | Higher solid waste volume, minimal wastewater erlab |
| Energy impact | Low when clean; risk of higher pressure drop if washing delayed sciencedirect | Predictable pressure drop curve; allows optimized changeout for energy use ph.parker |
| GMP complexity | Requires cleaning validation and reuse criteria erlab | Simpler documentation and risk assessment ph.parker |
| Best use cases | High‑dust, low‑risk zones; utility areas; technical corridors erlab | Pre‑HEPA in classified rooms; multi‑product and cytotoxic areas ph.parker |
Practical Implementation Steps for Pharma Facilities
7-Step Roadmap for Your Filtration Upgrade
1. Map your air streams across manufacturing, labs, utilities, and support areas, including sterilization cabinets and solution preparation rooms. [erlab]
2. Classify risk levels for each zone, considering product type, cytotoxicity, and cleanliness classification. [cleanroomworld]
3. Select filter roles: washable pre‑filters in low‑risk high‑dust paths, disposable synthetic media before HEPA in higher‑grade areas. [ph.parker]
4. Define monitoring points for pressure drop, airflow, and particle counts to build a data‑driven maintenance plan. [sciencedirect]
5. Validate cleaning SOPs for washable filters, including detergents, drying methods, and acceptance limits. [erlab]
6. Qualify suppliers of synthetic media for consistency, traceability, and documentation that supports GMP audits. [purolatorairfilters]
7. Review yearly to refine washing intervals and changeout thresholds based on actual performance and energy data. [sciencedirect]
For Ningbo Everheal's integrated production lines, embedding this roadmap into factory layout planning and HVAC design can differentiate your turnkey solution versus equipment‑only competitors. [syntegon.com]
Case Insight: Sustainable Air Filtration in Modern Labs
Industry case studies show that optimized air filtration can cut both environmental impact and operating costs in labs. Hospitals and universities using advanced filtration (HEPA plus optimized pre‑filters) have reported particle reductions of around 46–55% in sensitive rooms while lowering energy consumption. [erlab]
Similarly, labs that replaced traditional high‑flow exhaust systems with smarter filtered recirculation and efficient filters have achieved roughly 10% energy savings while improving user safety. Although each facility is unique, these examples confirm that data‑driven filtration strategies pay off in cleaner air, lower emissions, and better budgets. [mygreenlab]
Integrating Filtration Strategy with Sterilization and Solution Preparation Lines
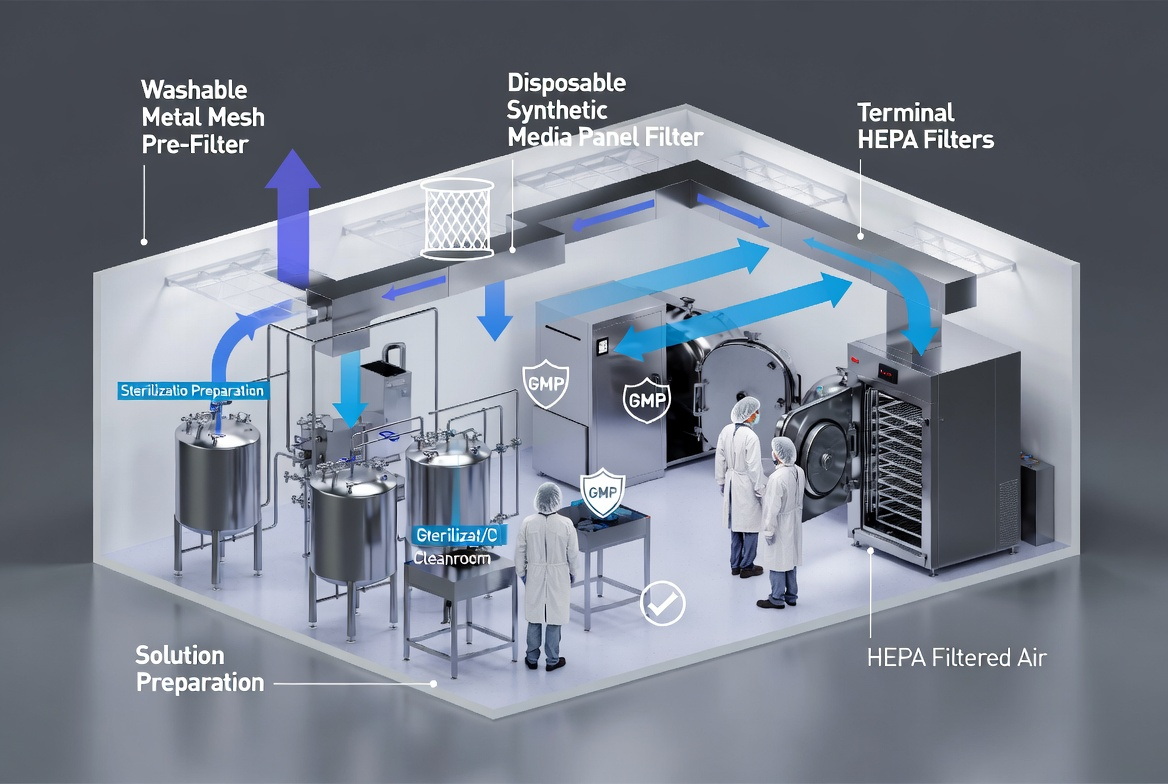
For a manufacturer of sterilization cabinets and pharmaceutical solution preparation systems, filter strategy must align with process risks, not just building HVAC. Areas such as mixing tanks for large‑volume parenterals, lyophilization loading/unloading, and anticancer drug preparation are especially sensitive to particulate and microbial control. [getinge]
In these zones, disposable synthetic pre‑filters plus high‑grade HEPA and controlled airflow patterns are typically essential, while washable pre‑filters can serve upstream to reduce dust loads and extend fine filter life. By designing air handling, layouts, and filtration together, you can deliver a turnkey cleanroom ecosystem rather than isolated equipment. [directindustry]
When Should You Choose Which? Expert Recommendations
From an expert standpoint, the choice is not either–or but where and how to use each technology. [sciencedirect]
Use washable pre‑filters when:
- Dust loads are high but contamination risk is moderate.
- You have reliable maintenance resources and can validate cleaning SOPs.
- Sustainability goals prioritize waste reduction on non‑critical air paths.
Use disposable synthetic media when:
- Air feeds cleanrooms, critical labs, or aseptic processing zones.
- You manage multiple high‑potency or cytotoxic products.
- Audit readiness and changeout simplicity are high priorities.
For many pharma plants, a data‑driven hybrid strategy delivers the best balance of sustainability, compliance, and cost. [sciencedirect]
Clear Call to Action (CTA)
If you are planning a new sterile manufacturing line or upgrading an existing high‑dust lab, integrate your filtration strategy into the early layout and HVAC design rather than treating it as an afterthought. Reach out to Ningbo Everheal's engineering team to discuss a tailored combination of washable pre‑filters and disposable synthetic media that aligns with your environmental goals, GMP profile, and long‑term operating costs. [erlab]
FAQs: Washable Pre-Filters vs. Disposable Synthetic Media
Q1. Are washable pre-filters suitable for GMP Grade A/B areas?
In most facilities, washable pre‑filters are used upstream in lower‑risk zones, while Grade A/B areas rely on disposable pre‑filters plus HEPA to simplify validation and avoid reuse‑related contamination risks. [cleanroomworld]
Q2. How often should disposable synthetic media be changed in high-dust labs?
Changeout frequency should be based on pressure‑drop trends, dust generation rate, and risk classification, but many labs define thresholds that trigger replacement before the maximum rated pressure drop. [ph.parker]
Q3. Do washable pre-filters always reduce operating costs?
They can reduce filter purchase and waste costs, but if cleaning is labor‑intensive or poorly scheduled, any savings may be offset by higher energy use or increased risk of variability. [directindustry]
Q4. Can filter choices impact regulatory inspections?
Yes. Regulators expect clear justification for where reusable filters are used, validated cleaning SOPs, and robust change‑control; disposable synthetic media can simplify documentation in high‑risk areas. [purolatorairfilters]
Q5. How does filtration tie into sterilization cabinet and solution preparation system design?
Air filtration defines the cleanliness of the environment around sterilization cabinets, tanks, and lines, influencing bioburden control, particle levels, and overall sterility assurance strategy. [syntegon.com]
References
1. Erlab – "Reducing the environmental impact of laboratories through air filtration." [erlab]
2. Parker – "Disposable Panel Filters with Synthetic Media (P312)." [ph.parker]
3. Purolator Air Filters – "P312 | Synthetic Panel Filters." [purolatorairfilters]
4. DirectIndustry – "Washable filter, Washable pre-filter – All industrial manufacturers." [directindustry]
5. Cleanroom World – "HEPA Filters | Filters for Cleanrooms." [cleanroomworld]
6. ScienceDirect – "Strategies for enhancing performance sustainability of air filtration systems." [sciencedirect]
7. My Green Lab – "Sustainable labs in action: Revolutionizing air filtration for greener research and industry." [mygreenlab]
8. Getinge – "GSS P Steam Sterilizer for pharmaceutical production." [getinge]
9. Syntegon – "Pharmaceutical Formulation and Processing Solutions." [syntegon.com]






















































