Content Menu
● What Is a Banana Bag?
● The Role of Infusion Bag Technology
● How Quickly Can Banana Bag Be Infused?
>> Understanding Infusion Rates
>> Why Rate Control Matters
● Factors Influencing Banana Bag Infusion Speed
>> 1. Clinical Condition
>> 2. Infusion Bag Material and Design
>> 3. Infusion System Configuration
>> 4. Solution Viscosity and Composition
>> 5. Environmental Conditions
● Manufacturing Perspective: Precision in Infusion Bag Production
>> Key Production Processes
● Infusion Bag System Optimization in Hospital Use
>> Benefits of Optimized Infusion Systems:
● Engineering Innovations by Everheal
● Clinical Guidelines for Safe Banana Bag Infusion
>> Recommended Guidelines:
● Technological Trends in Infusion Bag Systems
● Conclusion
● FAQ
>> 1. How quickly can a 1-liter Banana Bag be infused?
>> 2. What is inside a Banana Bag?
>> 3. Can Banana Bags be infused faster in emergencies?
>> 4. What kind of Infusion Bag is used for Banana Bags?
>> 5. How does Everheal support Infusion Bag production?
In modern medical treatment and pharmaceutical manufacturing, Infusion Bags play a central role in delivering vital nutrients, electrolytes, and medications safely and efficiently. Among these, the Banana Bag—a yellow-colored intravenous (IV) solution commonly used to treat dehydration or vitamin deficiencies—raises an important question for clinicians and healthcare engineers alike: how quickly can it be infused?
Understanding the infusion time of Banana Bags is crucial not only for healthcare providers but also for manufacturers and process engineers who design sterile infusion systems. This article explores infusion dynamics, key factors influencing flow rates, manufacturing considerations, and the technical aspects of Infusion Bag systems within pharmaceutical and hospital settings.

What Is a Banana Bag?
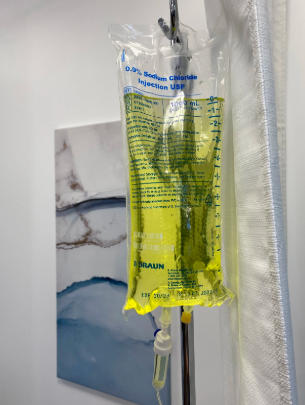
A Banana Bag, also known as a Multivitamin Infusion Bag, is an intravenous fluid that contains a mixture of vitamins and minerals, typically used for patients suffering from chronic alcohol use, malnutrition, or prolonged dehydration. The term “banana” comes from its bright yellow color, caused primarily by the presence of multivitamins, especially vitamin B2 (riboflavin).
A standard Banana Bag formula includes:
- 1 liter of 0.9% sodium chloride or dextrose solution
- 100 mg of thiamine (vitamin B1)
- 1 mg of folic acid
- 1 ampoule of multivitamin infusion (which provides the yellow tint)
Sometimes, magnesium sulfate or other components may be added, depending on the treatment protocol.
These solutions are stored in sterile Infusion Bags manufactured under strict Good Manufacturing Practice (GMP) standards, ensuring product purity and patient safety.
The Role of Infusion Bag Technology
Modern Infusion Bag technology is a cornerstone of both clinical therapy and pharmaceutical production. At companies like Everheal, advanced systems are designed to ensure bags remain sterile, durable, and compliant with international standards (such as USP and EP). The technology used in these systems influences how safely and efficiently Banana Bags and other IV fluids can be infused.
Key process systems include:
- Pure water preparation systems for producing Water for Injection (WFI)
- Pure steam generators used for sterilization
- Liquid filling and sealing machines for closed-system bag production
- Sterilization systems that eliminate microbial contamination
These integrated systems guarantee that every Infusion Bag maintains its integrity—critical for precise infusion rates and clinical reliability.
How Quickly Can Banana Bag Be Infused?
Understanding Infusion Rates
The time required to infuse a Banana Bag depends on multiple clinical and physical factors. Typically, a 1-liter Banana Bag is infused over 1 to 2 hours. However, infusion speed must be individualized based on the patient's condition, age, and medical history.
Typical Banana Bag Infusion Rates:
- Standard adult rate: 500–1000 mL/hour
- Slow maintenance infusion (elderly or compromised patients): 250–500 mL/hour
- Rapid correction (severe dehydration or emergency): up to 1500 mL/hour (under monitoring)
Why Rate Control Matters
Administering a Banana Bag too quickly can overload the cardiovascular system, leading to fluid imbalance, pulmonary edema, or electrolyte disturbances. Hence, infusion controllers, volumetric pumps, and specially designed Infusion Bags with pressure-resistant seams are used to maintain a safe and steady flow.
In industrial terms, the design of an Infusion Bag affects laminar flow, oxygen permeability, and pressure equilibria—all influencing how quickly the fluid can be infused without compromising quality.
Factors Influencing Banana Bag Infusion Speed
Several elements determine infusion speed, classified broadly into clinical, device-related, and environmental factors.
1. Clinical Condition
Patients with cardiac or renal dysfunction require slower infusion to prevent volume overload. Conversely, patients with severe dehydration may receive infusions faster to restore fluid balance rapidly.
2. Infusion Bag Material and Design
The physical properties of an Infusion Bag—including film thickness, flexibility, and internal pressure resistance—affect fluid flow consistency. Typically, Banana Bags are made from non-PVC multilayer film that maintains solution clarity and resists leaching.
3. Infusion System Configuration
A well-calibrated IV set or infusion pump controls the flow rate precisely. The inner diameter of the tubing, height differential between the Infusion Bag and patient, and any inline filters will alter the actual infusion time.
4. Solution Viscosity and Composition
A Banana Bag has slightly higher viscosity than normal saline due to added multivitamins and electrolytes. This can slow infusion rates by 5–10%, requiring minor adjustments in flow control.
5. Environmental Conditions
Temperature also affects fluid flow. Warm fluids may flow slightly faster compared to chilled solutions because viscosity decreases with heat.
Manufacturing Perspective: Precision in Infusion Bag Production
From a pharmaceutical engineering viewpoint, the manufacturing precision of an Infusion Bag directly impacts clinical performance, including infusion rate stability and aseptic integrity.
Key Production Processes
1. Water and Steam Purification
- Systems for pure water generation ensure the base solution meets pharmacopeial standards.
- Pure steam is used to sterilize bags, pipelines, and connectors.
2. Multi-effect Distillation Units
- Used to produce Water for Injection (WFI), providing an uncontaminated solvent for Banana Bag solutions.
3. Filling and Sealing
- Liquid filling machines insert precise dosages of saline and vitamins into each Infusion Bag.
- Hot sealing or ultrasonic welding ensures leak-proof seams.
4. Sterilization and Leak Detection
- Final autoclaving guarantees sterility.
- Leak-detection systems verify bag integrity and uniform pressure resistance.
These rigorous steps ensure that every Banana Bag produced is safe, stable, and ready for precise infusion control.

Infusion Bag System Optimization in Hospital Use
Hospitals are increasingly adopting closed-system infusion technologies, which prevent contamination and reduce exposure risk. These systems use sterile connectors and automatic flow regulators designed around infusion bag geometry.
Benefits of Optimized Infusion Systems:
- Accurate medicine dosing
- Consistent flow rates
- Reduced contamination risk
- Less manual intervention by staff
- Compatibility with automated monitoring systems
Modern Infusion Bags designed by compliant manufacturers can maintain stability even under pressurized infusion conditions, allowing safe administration within 1–2 hours.
Engineering Innovations by Everheal
As a leading provider of pharmaceutical equipment systems, Everheal integrates customized Infusion Bag manufacturing solutions for global clients. The company's systems ensure every stage—from WFI generation to final autoclaving—meets GMP requirements and maintains efficiency in Banana Bag production.
Everheal's key equipment lines include:
- Pure water preparation systems for infusion solution compounding
- Pure steam generators for aseptic production
- Multi-effect distillers for large-scale WFI generation
- Liquid filling and sealing equipment tailored for Infusion Bag production
- Sterilization and leak testing systems for quality assurance
Through these technologies, Everheal enables pharmaceutical firms and hospital pharmacy departments to produce Banana Bags that deliver precise composition control and safe infusion speed.
Clinical Guidelines for Safe Banana Bag Infusion
When infusing a Banana Bag, medical staff should follow protocol-driven procedures to ensure patient safety and optimize treatment outcomes.
Recommended Guidelines:
- Verify patient condition and adjust rate accordingly.
- Use IV infusion pumps for accurate flow regulation.
- Monitor for allergic reactions or infusion-related discomfort.
- Avoid mixing Banana Bag components directly in the same line with incompatible drugs.
- Document infusion duration for traceability.
Using approved Infusion Bags with ISO-standard connectors minimizes the risk of leaks, contamination, or dosing errors.
Technological Trends in Infusion Bag Systems
The pharmaceutical industry continues to evolve, introducing smarter and safer Infusion Bag technologies. Current innovation trends include:
- Non-PVC biocomposite materials for enhanced sustainability.
- Dual-chamber bags enabling on-demand mixing of vitamins and saline.
- Electronic RFID tagging for batch traceability.
- Automated filling systems ensuring dose accuracy within ±0.5%.
- Reduced-leach barrier films maintaining vitamin potency.
These trends highlight the growing convergence between biocompatible materials and digital process control in the Infusion Bag sector.
Conclusion
The infusion speed of a Banana Bag typically ranges between 1 and 2 hours, but it ultimately depends on patient needs, device calibration, and infusion bag quality. From a clinical perspective, rate control ensures safety and therapeutic precision. From a manufacturing and engineering standpoint, infusion bag design, sterilization systems, and production accuracy ensure reliability and compliance.
Companies like Everheal provide the technological backbone that supports the safe and effective use of Infusion Bags across the globe. Through integrated system design—from water preparation to sterilization—Everheal helps pharmaceutical clients deliver quality-assured solutions that combine clinical safety with engineering excellence.
FAQ
1. How quickly can a 1-liter Banana Bag be infused?
The standard infusion time is 1–2 hours, depending on the patient's condition and clinical judgment. Using an infusion pump with calibrated control helps regulate flow accurately.
2. What is inside a Banana Bag?
It typically contains saline or dextrose solution, thiamine (vitamin B1), folic acid, and multivitamins. Some formulations also include magnesium sulfate or additional electrolytes.
3. Can Banana Bags be infused faster in emergencies?
In severe dehydration or alcohol withdrawal emergencies, infusion may be faster—up to 1.5 liters per hour—but must always be supervised by medical professionals.
4. What kind of Infusion Bag is used for Banana Bags?
They are usually made of latex-free, non-PVC multilayer film bags to ensure chemical stability and prevent contamination.
5. How does Everheal support Infusion Bag production?
Everheal provides end-to-end pharmaceutical system solutions, including pure water systems, distillation equipment, liquid filling and sealing machines, and sterilization units tailored for safe and efficient Infusion Bag production.





















































