Content Menu
● Understanding the Role of the Infusion Bag
● Optimal Blood Storage Temperature
>> Why Blood is Stored Cold
● Are Blood Bags Cold When Infused?
>> Why Cold Infusion Can Be a Concern
● Blood Warmers and Infusion Systems
>> Key Features of a Blood Warming System
● Preparing Blood Bags for Transfusion
>> Step-by-Step Overview
● Infusion Bag Material and Safety Standards
>> Common Infusion Bag Materials
● The Role of Sterilization in Infusion Bag Production
● Advanced Manufacturing and Customization
● Clinical Considerations When Infusing Blood
● Benefits of Controlled Infusion Temperature
● Common Myths About Blood Infusion Temperature
● Conclusion
● FAQ
>> 1. What temperature should blood bags be stored at?
>> 2. Can you infuse blood directly from cold storage?
>> 3. How long does a blood Infusion Bag last?
>> 4. Are Infusion Bags reusable?
>> 5. How are Infusion Bags sterilized during production?
Blood transfusion is a critically important medical procedure in hospitals and clinical settings worldwide. Whether it's used to treat trauma patients, surgical cases, or chronic anemia, the process of transfusing blood must be handled with extreme precision to ensure patient safety. One frequently asked question is: are blood bags cold when infused?
To answer this question, it's essential to understand how Infusion Bags, particularly blood bags, are stored, prepared, and administered. This article explores every aspect of the topic — from the science of temperature control in transfusions to the advanced equipment used in modern healthcare facilities.
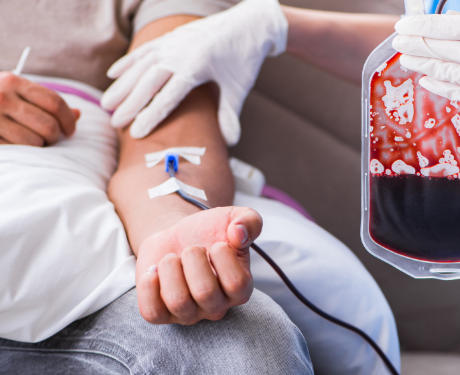
Understanding the Role of the Infusion Bag
An Infusion Bag is a sterile flexible container used to store and administer fluids, medications, nutritional solutions, or blood products to patients through an intravenous (IV) line. In the case of blood transfusions, these bags must maintain the viability of red blood cells, platelets, and plasma during both storage and administration.
Blood bags are a specialized type of Infusion Bag made of medical-grade PVC (polyvinyl chloride) or non-PVC materials. These materials must meet rigorous standards to avoid chemical interactions that could alter the integrity of blood components.
Typically, Infusion Bags are available in single or multiple chambers and can be designed for various medical purposes including:
- Intravenous saline or glucose administration
- Delivery of antibiotics and chemotherapy drugs
- Parenteral nutrition for patients unable to eat or absorb nutrients normally
- Blood storage and transfusion in hospital wards and emergency departments
Optimal Blood Storage Temperature
Blood safety depends largely on proper temperature regulation. Whole blood and red blood cell concentrates are stored at 2–6°C (35.6–42.8°F). This temperature range minimizes bacterial growth and preserves cell function.
However, blood stored in these conditions feels *cold* to the touch, which raises concerns about whether it should be infused directly into the patient without warming.
Why Blood is Stored Cold
Cold storage slows down metabolic processes in the cells, preventing premature degradation of red blood cells and hemoglobin. The same principle applies to vaccines and many injectable biological products. When blood is removed from its storage environment, it must be handled quickly — exposure to higher temperatures for too long can reduce shelf life and compromise sterility.
Are Blood Bags Cold When Infused?
In most cases, blood bags are slightly cold when infused, especially for small transfusion volumes or routine procedures. The infusion process typically occurs quickly enough that the mild coldness does not cause any harm.
However, for large transfusions, rapid transfusions, or vulnerable patients (such as infants, elderly individuals, or those undergoing surgery), blood should be warmed to approximately body temperature (37°C or 98.6°F) before infusion.
Why Cold Infusion Can Be a Concern
Infusing cold blood directly into the bloodstream can lead to:
- Hypothermia: A drop in body temperature, especially if large volumes are given.
- Cardiac stress: Cold blood can cause vasoconstriction, slowing circulation and increasing cardiac workload.
- Shivering and discomfort: Even mild temperature differences can cause muscular reactions.
Thus, modern hospitals use blood-warming devices — devices compatible with Infusion Bag systems — to ensure patient comfort and safety.
Blood Warmers and Infusion Systems
A blood warmer is an electrical or water-based device that gently raises the temperature of blood or IV fluids before infusion. It ensures that the Infusion Bag's contents enter the patient's bloodstream at a safe, physiologically compatible temperature.
Key Features of a Blood Warming System
- Temperature control sensors: Maintain precise ranges, typically 35–42°C.
- High flow compatibility: Especially important for rapid transfusion systems.
- Alarm functions: Alert operators if blood temperature deviates from the set range.
- Compatibility: Can be connected to most Infusion Bags or integrated within automated infusion systems.
Everheal, as a manufacturer specializing in pharmaceutical and medical process equipment, provides comprehensive production line solutions including sterilization systems, pure steam generators, and Infusion Bag assembly solutions. Such high-standard equipment ensures that every bag used in healthcare meets strict sterilization and regulatory requirements.
Preparing Blood Bags for Transfusion
The preparation process ensures that both the Infusion Bag and its contents are in optimal condition for administration.
Step-by-Step Overview
1. Verification and Identification:
Blood type, donor information, and expiration date are double-checked to avoid mismatches.
2. Inspection:
Bags are visually inspected for clots, discoloration, or signs of contamination.
3. Temperature Equilibration:
The cold Infusion Bag is sometimes allowed to reach room temperature for a brief period before warming or direct administration, depending on protocol.
4. Connection to IV Set:
The sterile infusion set is attached under aseptic conditions. The system is primed to remove air bubbles.
5. Infusion:
The process begins under supervision, and patient vitals are monitored throughout to ensure stable hemodynamics.
Infusion Bag Material and Safety Standards
The safety and performance of any Infusion Bag rely heavily on the raw material. Medical-grade materials must comply with global standards such as USP Class VI, ISO 8536, and EU Pharmacopoeia requirements.
Common Infusion Bag Materials
- PVC (Polyvinyl Chloride):
Durable and cost-effective, suitable for most pharmaceutical fluids.
- Non-PVC (Polypropylene or Polyolefin):
Ideal for sensitive biological preparations, free of plasticizers like DEHP that could leach into fluids.
- Multilayer films:
Provide improved oxygen, moisture, and gas barrier performance.
Safety testing includes assessments for extractables, leachables, transparency, flexibility, and sterilization resistance. Manufacturers like Everheal optimize film formulations to ensure product reliability in large-scale pharmaceutical applications.
The Role of Sterilization in Infusion Bag Production
Sterility is paramount, particularly for blood and injectable solutions. During Infusion Bag production, sterilization can be achieved through various methods:
- Autoclaving: Using saturated steam at 121°C for 15–20 minutes.
- Gamma irradiation: Often applied for non-heat-resistant materials.
- Ethylene oxide (EtO): Effective for delicate polymer films.
Everheal's sterilization systems are designed to ensure full sterilant penetration into packaging materials and complex piping systems, guaranteeing cleanliness throughout the production line.
Advanced Manufacturing and Customization
Modern hospitals increasingly demand customized Infusion Bag systems that integrate smart monitoring, labeling, and automation. Everheal's advanced manufacturing solutions allow pharmaceutical clients to design end-to-end sterile liquid filling, sealing, and packaging equipment suited for various applications — from saline and glucose bags to specialized infusion therapies.
Typical production systems include:
- Purified water preparation systems
- Pure steam generators
- Multi-effect distillation water machines
- Liquid filling and sealing lines
- Sterilization and cleanroom control systems
Such integration ensures optimal hygiene, consistency, and process efficiency — all vital factors for global healthcare compliance.
Clinical Considerations When Infusing Blood
Healthcare professionals consider several physiological factors before initiating a transfusion. These include:
- Patient's body temperature
- Volume and speed of infusion
- Underlying medical conditions
- Environmental temperature in the operating room or ward
If multiple Infusion Bags are connected sequentially, the system ensures stable temperature control across processes. For example, in massive transfusions, a series of pre-warmed bags may be prepared using continuous warming devices to reduce hypothermia risk.
Benefits of Controlled Infusion Temperature
Maintaining an optimal infusion temperature benefits patients by:
- Preventing thermal shock to the circulatory system
- Improving red cell survival rates post-transfusion
- Enhancing patient comfort during long-duration infusions
- Reducing recovery time for surgical or trauma patients
This shows that temperature-controlled Infusion Bags are essential components of modern transfusion systems, bridging patient safety with advanced biomedical engineering.
Common Myths About Blood Infusion Temperature
1. “Cold blood transfusions are dangerous.”
- Not always. Small volumes can be infused cold without major risks if monitored properly.
2. “Blood must always be warmed.”
- Only necessary for rapid or large-volume transfusions, or when patients have temperature vulnerabilities.
3. “Infusion Bags can be warmed in hot water manually.”
- False. Improper warming may damage cells. Only approved warming devices should be used.
4. “Storage and transfusion temperatures are the same.”
- No. Storage ensures preservation, while transfusion focuses on safe physiological compatibility.
Understanding these points helps clinicians adhere to best practices in patient care.
Conclusion
Blood bags, as specialized Infusion Bags, are generally stored cold at 2–6°C to preserve red cell integrity. Whether they remain cold when infused depends on clinical context. For standard transfusions, mild cold is acceptable; however, for rapid or massive infusions, warming is essential to maintain patient stability.
Modern healthcare relies on advanced Infusion Bag technology, precision sterilization, and controlled production systems — all areas where companies like Everheal provide comprehensive solutions. Through proper handling, manufacturing, and medical protocols, infusion safety and efficiency can be optimized for global patients.
FAQ
1. What temperature should blood bags be stored at?
Blood bags must be stored between 2–6°C for optimal preservation of red blood cells and to prevent bacterial growth.
2. Can you infuse blood directly from cold storage?
Yes, small transfusions can be infused directly if the temperature is within safe limits. However, for large transfusions or sensitive patients, the Infusion Bag should be warmed.
3. How long does a blood Infusion Bag last?
Typically, whole blood has a shelf life of 35–42 days, depending on additive solutions and storage conditions.
4. Are Infusion Bags reusable?
No. Infusion Bags are single-use sterile devices to prevent cross-contamination and maintain medical hygiene standards.
5. How are Infusion Bags sterilized during production?
They are sterilized using autoclaving, gamma irradiation, or ethylene oxide gas to ensure full microbial elimination without damaging material integrity.





















































