Content Menu
● Understanding the Purpose of a Pressure Bag
● How a Pressure Bag Influences Infusion Speed
● Typical IV Infusion Rates with Pressure Bags
● Factors Affecting IV Infusion Speed
>> 1. Pressure Applied
>> 2. Diameter and Length of Tubing
>> 3. Fluid Viscosity
>> 4. Altitude and Gravity
>> 5. Equipment Quality
● Applications of Pressure Infusion Systems
● Designing an Effective Pressure Infusion Setup
● Safety Considerations and Precautions
● Role of Everheal in Pharmaceutical Infusion System Design
● Conclusion
● FAQs
>> (1) How much pressure should a pressure bag apply during infusion?
>> (2) Can a pressure bag damage the infusion bag?
>> (3) How long does it take to infuse 1 liter using a pressure bag?
>> (4) Is a pressure bag necessary for blood transfusions?
>> (5) Can infusion speed be calculated manually?
Intravenous (IV) therapy is one of the most common and essential medical procedures used worldwide. It's a method for delivering fluids, medications, and nutrients directly into a patient's circulatory system. The infusion bag plays a central role in this process, and the rate of infusion is often controlled by gravity or by external devices such as a pressure bag. Understanding how fast IV fluid infuses when aided by pressure is critical for healthcare professionals to ensure patient safety and treatment efficiency.
This article explores the science behind IV infusion using pressure bags, including key influencing factors, calculation methods, and real-world applications—providing clarity for clinical users, engineers designing infusion systems, and pharmaceutical manufacturers alike.
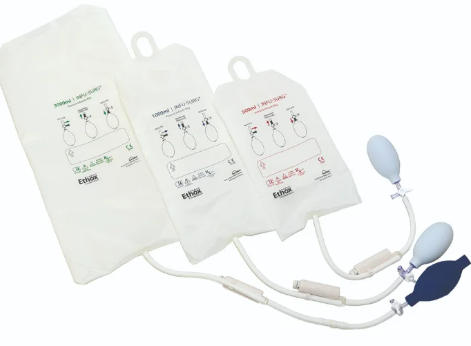
Understanding the Purpose of a Pressure Bag
A pressure bag is a medical device used to apply external pressure to an infusion bag containing IV fluids. By inflating the pressure bag around the fluid container, medical staff can increase the fluid delivery rate, ensuring rapid administration of fluids in emergency or critical care situations.
Normally, IV fluids rely on gravitational force—the higher the IV bag, the faster the fluid flows. However, in cases such as severe dehydration, trauma, or shock, time is critical and gravity alone may not provide an adequate infusion rate. Here, the pressure infusion bag becomes invaluable, enabling clinicians to deliver fluids at a controlled and accelerated rate.
How a Pressure Bag Influences Infusion Speed
The speed at which an IV fluid infuses under pressure depends on multiple variables. The most important one is the amount of pressure applied by the pressure bag—typically measured in millimeters of mercury (mmHg). A usual operating range for infusion pressure bags is between 150 to 300 mmHg.
In general, greater pressure leads to faster infusion. The relationship between infusion speed and applied pressure can be explained through Poiseuille's Law, which defines flow rate through a cylindrical tube:
Q=π⋅r4⋅ΔP/8⋅μ⋅L
Where:
QQ = flow rate,
rr = radius of the tubing,
ΔPΔP = pressure difference,
μμ = fluid viscosity,
LL = tubing length.
From this formula, we see that flow rate increases with higher pressure (\(\Delta P\)) and larger tubing diameters (\(r\)), but decreases with higher viscosity (\(\mu\)) or longer tubing (\(L\)).
Typical IV Infusion Rates with Pressure Bags
Under normal gravity, a typical infusion bag might deliver fluids at a rate of 50–150 mL per minute depending on bag height and tubing specifications. When a pressure bag of 300 mmHg is applied, the flow can increase significantly, often reaching rates of 300–500 mL per minute or even higher, depending on system configuration.
Here are some typical scenarios:
| Infusion Pressure (mmHg) | Approximate Flow Rate (mL/min) | Common Clinical Application |
| 0 (Gravity only) | 50–150 | Routine IV administration |
| 100 | 200–300 | Moderate fluid resuscitation |
| 200 | 350–450 | Rapid fluid delivery |
| 300 | 400–600 | Emergency trauma care |
Clinically, a 1-liter infusion bag can be completely infused in 2–3 minutes using a 300 mmHg pressure bag, compared with 10–15 minutes using gravity flow.
Factors Affecting IV Infusion Speed
Several factors govern how fast an IV fluid can infuse under pressure:
1. Pressure Applied
The level of pressure applied directly affects the infusion rate. A precise balance must be maintained—too little pressure slows infusion, while excessive pressure may rupture the infusion bag or cause vein infiltration.
2. Diameter and Length of Tubing
A wide-bore IV catheter (e.g., 14G or 16G) allows fluid to pass more quickly than a narrower catheter. Shorter tubing also minimizes flow resistance, improving efficiency.
3. Fluid Viscosity
Thicker fluids such as blood products or nutrient solutions flow more slowly compared to crystalloids (like normal saline). The infusion bag composition and temperature also influence viscosity.
4. Altitude and Gravity
Environmental pressure and local gravity subtly affect the differential pressure between the infusion bag and vein, which can influence flow to a small extent.
5. Equipment Quality
High-quality infusion bags designed with strong materials, precise seals, and uniform wall thickness maintain consistent pressure distribution—minimizing risk of leaks or irregular infusions. In contrast, low-quality bags may deform or rupture under high pressure.
Applications of Pressure Infusion Systems
Pressure-based infusion systems are widely used across clinical fields and pharmaceutical operations:
- Emergency and trauma care: Rapid fluid replacement is often life-saving.
- Operating rooms: Maintaining blood pressure and hydration levels during anesthesia.
- Critical care units: Delivering precise medication dosages or total parenteral nutrition.
- Pharmaceutical manufacturing: Pressure testing and performance validation of infusion bags for product reliability and compliance.
For manufacturers like Everheal, which specializes in sterilization systems, distilled water generators, and pharmaceutical-grade infusion bag solutions, optimizing design and performance testing of pressure infusion systems ensures global compliance with GMP and FDA standards.
Designing an Effective Pressure Infusion Setup
For healthcare engineers and clinical managers, designing a reliable infusion setup involves integrating several technical aspects:
1. Material Selection: Choose medical-grade PVC or multilayer polymer films for the infusion bag to withstand up to 300 mmHg safely.
2. Pressure Control: Equip pressure bags with calibrated gauges for accuracy.
3. Leak Testing: Conduct hydrostatic pressure tests to validate bag durability.
4. Sterility Assurance: Use steam sterilization or gamma irradiation systems to maintain aseptic integrity.
5. Automation Integration: For industrial-scale applications, automation can control infusion speed, maintain pressure consistency, and ensure traceability.
By combining precision-engineered infusion bags with advanced sterilization and monitoring systems, manufacturers can enhance performance, safety, and compliance simultaneously.
Safety Considerations and Precautions
While pressure infusion provides speed and efficiency, safety remains paramount. Improper pressure management can cause complications such as vessel damage or bag rupture. Here are key safety measures:
- Monitor pressure regularly: Never exceed 300 mmHg unless specifically indicated.
- Avoid air entrapment: Always remove air bubbles before connecting the infusion bag.
- Inspect equipment: Ensure both the IV catheter and tubing connections are tight and undamaged.
- Follow manufacturer instructions: Use only approved pressure devices compatible with the IV system.
- Observe patient response: Monitor for signs of discomfort, swelling, or infusion site leakage.
Role of Everheal in Pharmaceutical Infusion System Design
As a global supplier of pharmaceutical equipment, Everheal provides comprehensive solutions including pure water systems, pure steam generators, multi-effect distilled water machines, liquid filling and sealing machines, and advanced sterilization systems. These technologies help pharmaceutical manufacturers build integrated production lines for sterile IV fluids and infusion bags.
Our engineering expertise extends to the design of automated pressure testing and validation systems, ensuring each infusion bag can safely withstand rapid infusion conditions. We support clients in factory layout planning, equipment customization, and GMP-compliant production line installation—enabling efficient, high-quality pharmaceutical manufacturing.
By aligning advanced engineering with international safety standards, Everheal is helping the global medical industry deliver safe and efficient infusion products to hospitals and healthcare providers worldwide.
Conclusion
The rate at which an IV infuses by pressure bag depends on several factors including applied pressure, tubing size, fluid viscosity, and infusion bag quality. Generally, using a pressure bag significantly accelerates infusion—often enabling 1 liter of fluid to be infused within a few minutes under 300 mmHg.
For hospitals, this speed is vital in emergencies; for manufacturers, this performance ensures their infusion bag products meet clinical requirements. Proper design, testing, and compliance are necessary to ensure safety and consistent flow performance.
With advanced systems from companies like Everheal, pressure-assisted infusion technology continues to evolve—offering faster, safer, and more reliable fluid delivery worldwide.
FAQs
(1) How much pressure should a pressure bag apply during infusion?
Most standard protocols recommend 150–300 mmHg pressure for rapid infusions. Lower pressures are used for routine IV therapy, while higher pressures are limited to emergency scenarios.
(2) Can a pressure bag damage the infusion bag?
Not if high-quality infusion bags are used. Bags designed for medical use are tested for burst resistance. However, excessive inflation beyond the recommended pressure may risk leakage.
(3) How long does it take to infuse 1 liter using a pressure bag?
At approximately 300 mmHg, 1 liter of saline solution can infuse in 2–3 minutes, depending on tubing gauge and patient vein size.
(4) Is a pressure bag necessary for blood transfusions?
Yes, particularly during emergency transfusions or when large volumes are needed rapidly. However, temperature control and proper filtration are crucial to avoid hemolysis.
(5) Can infusion speed be calculated manually?
Yes. By using Poiseuille's equation and considering tubing size, viscosity, and pressure differences, clinicians or engineers can estimate infusion flow rates quite accurately.





















































