Content Menu
● What Is A Pressure Infusion Bag?
● Key Components You Need
● Step‑By‑Step: How To Use A Pressure Infusion Bag
>> Step 1: Verify the Infusion Bag
>> Step 2: Prepare the IV Line
>> Step 3: Insert the Infusion Bag Into the Pressure Cuff
>> Step 4: Hang the Assembly
>> Step 5: Inflate to the Target Pressure
>> Step 6: Connect to the Patient
>> Step 7: Monitor During Infusion
>> Step 8: Deflate and Remove After Use
● Safety Tips and Best Practices
>> Choose the Right Infusion Bag
>> Avoid Over‑Pressurization
>> Prevent Air Embolism
>> Maintain Sterility
● Integration With Modern Infusion Systems
● Common Mistakes To Avoid
● When Should You Use A Pressure Infusion Bag?
● Conclusion
● FAQ
>> 1. What is the safe pressure range for a pressure infusion bag?
>> 2. Can I reuse a pressure infusion bag?
>> 3. Is it safe to use a pressure infusion bag with an infusion pump?
>> 4. Can I use a pressure infusion bag for all types of Infusion Bags?
>> 5. What should I do if the Infusion Bag starts leaking under pressure?
In modern medical practice, a pressure infusion bag is an essential tool for rapid fluid delivery, blood transfusion, and controlled infusion in emergency and critical care. Whether in an ICU, operating room, ambulance, or general ward, correct use of a pressure infusion bag directly impacts infusion speed, patient safety, and medication accuracy.
This guide explains step by step how to use a pressure infusion bag, what to check before use, how to monitor during infusion, and how to avoid common mistakes. If your facility uses automated infusion systems, infusion pumps, or integrated Infusion Bag solutions, the same safety principles still apply.

What Is A Pressure Infusion Bag?
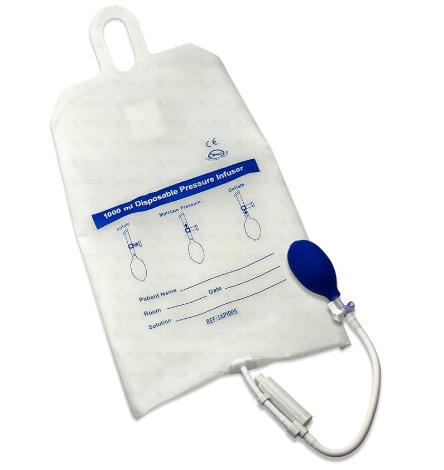
A pressure infusion bag is a reusable inflatable cuff designed to apply external pressure around an IV fluid bag, blood bag, or medication Infusion Bag. By inflating the cuff, you increase the pressure surrounding the fluid, which accelerates flow through the IV line.
Typical uses include:
- Rapid volume resuscitation in shock or trauma
- Accelerated blood and plasma transfusion
- Controlled infusion during anesthesia or surgery
- Infusion in situations where gravity alone is not enough (e.g., low-hanging bags, field environments)
The pressure infusion bag usually has:
- A transparent window to visualize the fluid level in the Infusion Bag
- A hand bulb (or pump) with a one-way valve to inflate the cuff
- A pressure gauge (often 0–300 mmHg)
- A pressure release valve or button to deflate after use
- Straps or a sleeve to hold the Infusion Bag in place
Key Components You Need
Before learning how to use a pressure infusion bag, confirm you have the complete setup:
- Sterile IV fluid or blood Infusion Bag (correct medication, volume, and concentration)
- Pressure infusion bag (correct size for your Infusion Bag, commonly 500 ml, 1000 ml, or 3000 ml)
- IV giving set (infusion tubing) with a drip chamber and roller clamp
- IV catheter inserted into the patient (appropriate gauge)
- IV pole or mounting hook
- Alcohol swabs, labels, and necessary documentation
If your hospital uses advanced systems like automated liquid filling and sealing lines or sterile Infusion Bag manufacturing solutions, the clinical handling steps for the filled Infusion Bag remain similar once it arrives at the bedside.
Step‑By‑Step: How To Use A Pressure Infusion Bag
Step 1: Verify the Infusion Bag
- Check the Infusion Bag label: drug name, concentration, volume, patient name (if pre-labeled), and expiration date.
- Inspect the Infusion Bag for leaks, cloudiness, particles, color changes, or damaged ports.
- Confirm compatibility with other medications and solutions the patient is receiving.
A pressure infusion bag should only be used on intact, undamaged Infusion Bags. Never apply pressure to a compromised bag; this can cause rupture and contamination.
Step 2: Prepare the IV Line
1. Open the IV set using aseptic technique.
2. Spike the Infusion Bag at the designated port without touching the spike tip.
3. Invert the Infusion Bag and gently squeeze to fill the drip chamber to the recommended level (about one‑third to one‑half full).
4. Prime the IV line completely to remove air. Adjust the roller clamp to stop flow until you are ready to connect to the patient.
Proper priming is critical. When a pressure infusion bag increases flow rate, any residual air can be pushed quickly into the patient's bloodstream, which is dangerous.
Step 3: Insert the Infusion Bag Into the Pressure Cuff
- Open the pressure infusion bag cuff fully.
- Place the Infusion Bag inside the sleeve with the ports facing downward and the label or fluid window facing outward.
- Align the Infusion Bag so you can easily see the fluid level through the viewing window.
- Secure any straps, clips, or closures so the Infusion Bag does not slide inside the cuff.
The Infusion Bag should sit flat and centered so pressure is applied evenly. Uneven pressure can distort the bag and affect your ability to read the volume accurately.
Step 4: Hang the Assembly
- Hang the pressure infusion bag with the Infusion Bag inside on an IV pole hook or wall bracket.
- Ensure the IV tubing hangs straight down without kinks or tight bends.
- Confirm that the patient's IV catheter site is lower than the Infusion Bag to support flow.
Even though the pressure infusion bag can drive fluid against gravity, proper positioning still improves performance and reduces alarms or interruptions.
Step 5: Inflate to the Target Pressure
- Close the pressure release valve on the hand bulb (if applicable).
- Start inflating by squeezing the hand bulb repeatedly.
- Watch the pressure gauge as the pressure infusion bag expands around the Infusion Bag.
Common target pressures:
- For most IV crystalloid Infusion Bags: \(150\text{–}300 \,\text{mmHg}\) depending on urgency and protocol.
- For blood Infusion Bags: typically around \(150\text{–}200 \,\text{mmHg}\), following your facility's policy.
Do not exceed the manufacturer's recommended pressure. Excessive pressure may damage the Infusion Bag, the ports, or increase hemolysis in blood products.
Step 6: Connect to the Patient
- Disinfect the IV catheter hub with an alcohol swab.
- Remove the protective cap from the primed IV line.
- Connect the line securely to the IV catheter or extension set.
- Open the roller clamp and observe the drip chamber to confirm flow.
If you see a rapid drip but poor patient response, confirm catheter patency, tubing integrity, and the position of the Infusion Bag within the pressure infusion bag.
Step 7: Monitor During Infusion
While the pressure infusion bag is in use, monitor:
- Patient status: vital signs, perfusion, consciousness level, and signs of fluid overload.
- Infusion site: redness, swelling, pain, or leakage (infiltration or extravasation).
- Infusion rate: drip rate and actual clinical response. A pressure infusion bag accelerates flow, so adjust treatment as necessary.
- Pressure gauge: maintain within the target range. Pressure will drop gradually as the Infusion Bag empties; you may need to reinflate.
Also track the volume infused. When the Infusion Bag is nearing empty, reduce the pressure to avoid pushing air into the line, then clamp and change bags safely.
Step 8: Deflate and Remove After Use
Once the Infusion Bag has finished:
- Close the roller clamp on the IV line.
- Open the pressure release valve on the pressure infusion bag to let air escape.
- Allow the cuff to deflate completely before removing the empty Infusion Bag.
- Disconnect and dispose of the used Infusion Bag and IV set according to your facility's infection control policy.
- Clean and disinfect the pressure infusion bag if it is reusable, following the manufacturer's instructions.
Never leave a pressure infusion bag inflated around an empty Infusion Bag. This can strain seams and shorten the equipment's lifespan.
Safety Tips and Best Practices
Choose the Right Infusion Bag
- Match Infusion Bag size to the pressure cuff size for better control.
- Verify that the Infusion Bag material (often PVC or non‑PVC for specific drugs) is compatible with pressure devices.
- For cytotoxic or high‑risk drugs, use closed system Infusion Bag solutions and follow specialized safety protocols.
Avoid Over‑Pressurization
- Always monitor the pressure gauge.
- Stay within recommended limits for crystalloids, colloids, and blood Infusion Bags.
- If you notice ballooning, bulging seams, or unusual deformation of the Infusion Bag, stop inflation and inspect for damage.
Prevent Air Embolism
- Prime the line completely before connecting.
- When the Infusion Bag is almost empty, gradually reduce cuff pressure.
- Do not allow the pressure infusion bag to squeeze a nearly empty Infusion Bag while the line is still open.
Maintain Sterility
- The pressure infusion bag itself is not sterile, but anything that touches the fluid pathway must remain sterile.
- Handle the ports and spike carefully when attaching the Infusion Bag and IV set.
- In high‑risk settings, consider sterile covers or drapes around the Infusion Bag area if required by protocol.
Integration With Modern Infusion Systems
In many hospitals, pressure infusion bags are used alongside:
- Infusion pumps and syringe pumps
- Automated infusion monitoring systems
- Pre‑filled, factory‑sealed Infusion Bag products produced on sterile filling and sealing equipment
When combining a pressure infusion bag with a pump, follow these principles:
- Confirm the pump is approved for use with pressurized Infusion Bags.
- Set the pressure low enough to avoid triggering pump alarms or causing occlusion.
- Use the pump's programmed rate to fine‑tune infusion speed, letting the pressure infusion bag provide basic driving pressure.
If your facility uses advanced filling, sealing, and sterilization solutions (e.g., automated liquid filling machines, steam sterilizers, purified water systems for producing Infusion Bag solutions), these technologies ensure that the Infusion Bag is safe and consistent before it reaches clinical use. The bedside nurse then applies the correct pressure infusion bag technique as the final step to deliver that sterile product to the patient.
Common Mistakes To Avoid
- Applying a pressure infusion bag to an Infusion Bag with visible damage or contamination
- Forgetting to prime the IV line completely before pressurizing
- Exceeding recommended pressure values and stressing the Infusion Bag
- Leaving the cuff inflated around an empty Infusion Bag
- Failing to monitor patient status while focusing only on the pressure gauge
- Using a pressure infusion bag in patients where rapid infusion is contraindicated (e.g., severe heart failure without close supervision)
When Should You Use A Pressure Infusion Bag?
Use a pressure infusion bag when:
- You need rapid fluid resuscitation and gravity infusion is too slow.
- The Infusion Bag cannot be hung high enough to produce adequate gravity pressure.
- Blood or fluid must be given quickly and safely in operating rooms, emergency rooms, ambulances, or field hospitals.
Avoid or use with extreme caution when:
- The patient has a high risk of fluid overload.
- The Infusion Bag contains drugs that must be delivered very slowly or require strict pump control.
- Institutional policy prohibits pressurized infusions for specific therapies.
Always follow local protocols, manufacturer guidelines for the pressure infusion bag, and the labeling on each Infusion Bag product.
Conclusion
A pressure infusion bag is a simple but powerful tool that can transform a standard Infusion Bag into a rapid, controlled delivery system for critical fluids and medications. By choosing the right Infusion Bag, setting up the IV line correctly, inflating only to safe pressures, and monitoring both the patient and the infusion system continuously, clinicians can achieve faster resuscitation with high safety and reliability.
In modern healthcare environments that rely on sterile Infusion Bag production, advanced water purification, steam generation, and automated filling technologies, correct bedside handling remains the last and most crucial link in the chain. Understanding how to use a pressure infusion bag properly ensures that the benefits of high‑quality Infusion Bag solutions are fully delivered to the patient when every second counts.
FAQ
1. What is the safe pressure range for a pressure infusion bag?
For most crystalloid Infusion Bags, a typical pressure range is \(150\text{–}300 \,\text{mmHg}\), depending on clinical urgency and hospital policy. For blood Infusion Bags, many guidelines recommend staying around \(150\text{–}200 \,\text{mmHg}\) to reduce the risk of hemolysis. Always check the instructions for both the pressure infusion bag and the specific Infusion Bag product you are using.
2. Can I reuse a pressure infusion bag?
Most pressure infusion bags are reusable devices, but they must be inspected, cleaned, and disinfected according to the manufacturer's instructions between patients. The Infusion Bag itself, however, is strictly single‑use. Never reuse an Infusion Bag or IV set, as this can cause cross‑infection and medication errors.
3. Is it safe to use a pressure infusion bag with an infusion pump?
It can be safe if both the pump and the pressure infusion bag are approved for combined use and you follow institutional guidelines. In such setups, the pressure infusion bag provides baseline pressure around the Infusion Bag, while the pump controls the exact flow rate. Keep the pressure low enough to avoid triggering occlusion alarms and always monitor both the pump and the patient.
4. Can I use a pressure infusion bag for all types of Infusion Bags?
No. Some medications or specialized Infusion Bags are not intended for use under pressure. Examples include certain chemotherapy drugs, highly viscous solutions, or Infusion Bags with fragile materials. Always read the Infusion Bag label and the drug's prescribing information. If in doubt, consult pharmacy or your institution's guidelines before applying a pressure infusion bag.
5. What should I do if the Infusion Bag starts leaking under pressure?
Stop the infusion immediately by closing the roller clamp and reducing the pressure in the pressure infusion bag. Remove the leaking Infusion Bag from the cuff, dispose of it safely according to your facility's hazardous waste procedures, and replace it with a new, intact Infusion Bag. Inspect the pressure infusion bag for sharp edges or damage that might have contributed to the leak, and do not use damaged equipment again.





















































